Number Of Protons For Gold
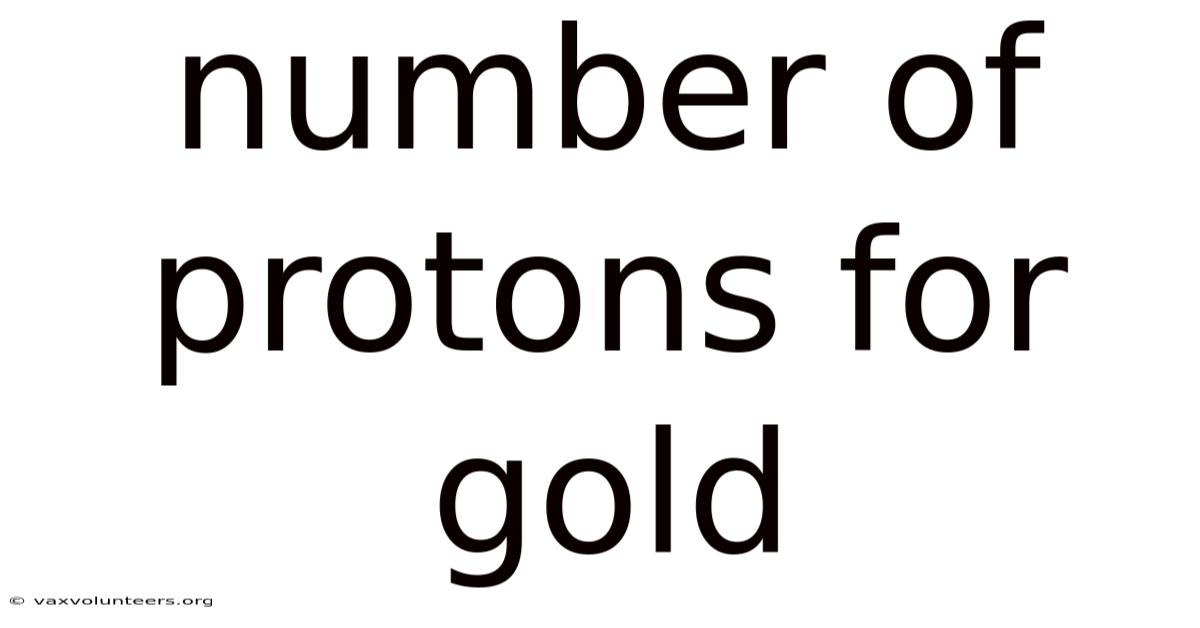
The Atomic Signature: Understanding the Number of Protons in Gold
At the heart of every atom lies a fundamental truth that defines its very identity: the number of protons in its nucleus. For the element gold, celebrated for its luster, rarity, and historical significance, this number is not just a trivial fact but the atomic cornerstone of its existence. The number of protons in a gold atom is 79. This single integer, known as the atomic number, is what makes gold gold and not any other element on the periodic table. It is the immutable, non-negotiable signature that separates gold from its neighbors, platinum (78 protons) and mercury (80 protons). Understanding this number unlocks a deeper comprehension of chemistry, physics, and the material world itself, revealing why gold possesses its unique color, density, and chemical inertness. This article will explore the profound implications of gold's 79 protons, moving from basic atomic theory to the sophisticated nuclear physics that explains its place in the universe.
Detailed Explanation: Protons, Atoms, and the Definition of an Element
To grasp the significance of gold having 79 protons, one must first understand the basic architecture of an atom. An atom consists of a tiny, dense nucleus at its center, surrounded by a cloud of electrons. The nucleus itself is composed of two types of particles: protons, which carry a positive electrical charge, and neutrons, which are electrically neutral. The number of protons in the nucleus is the atomic number (Z), and it is this number that determines the chemical identity of an element. Change the proton count, and you have a different element entirely—a process known as nuclear transmutation.
The electrons, which are negatively charged, orbit the nucleus in specific energy levels or shells. In a neutral atom, the number of electrons equals the number of protons. Therefore, a neutral gold atom also has 79 electrons. These electrons govern the atom's chemical behavior—how it bonds, reacts, and interacts with other atoms. The arrangement of these 79 electrons, particularly the single electron in its outermost shell, explains gold's reluctance to react with most substances, contributing to its famed resistance to corrosion and tarnish. Meanwhile, the 79 protons define the element's position on the periodic table (Group 11, Period 6) and its fundamental nuclear properties. The sum of protons and neutrons gives the mass number (A), which for the most common and stable isotope of gold is 197 (79 protons + 118 neutrons = 197). This isotope, gold-197, is what you find in jewelry, coins, and electronic components.
Step-by-Step Breakdown: Finding the Proton Count
Determining the number of protons for any element, including gold, follows a universal, logical process rooted in the organization of the periodic table.
- Locate the Element on the Periodic Table: Find the symbol for gold, which is Au (from the Latin aurum). It is situated in the sixth period and the eleventh group, often colored yellow or orange in many table designs.
- Identify the Atomic Number: The atomic number (Z) is always displayed as a whole number, typically centered or placed above the element's symbol on the periodic table. For gold, this number is 79.
- Understand the Direct Correlation: The atomic number is the number of protons. There is no calculation needed; it is a defined property. Therefore, every atom of gold has exactly 79 protons in its nucleus. This is an inviolable rule of chemistry.
- Distinguish from Mass Number: The number often shown below the symbol is the atomic mass (approximately 196.97 u for gold). This is a weighted average of all naturally occurring isotopes' masses. To find the neutron count in a specific isotope, you subtract the atomic number (protons) from the mass number (protons + neutrons). For gold-197: 197 - 79 = 118 neutrons.
This step-by-step method works for every element, making the periodic table an indispensable map to the building blocks of matter.
Real Examples: Why 79 Protons Matter in the Real World
The fact that gold has 79 protons is not merely academic; it has direct, tangible consequences that shape its role in technology, economics, and art.
- Chemical Inertness and Jewelry: The electron configuration resulting from 79 protons and 79 electrons makes gold's outermost shell almost full. This gives it an extremely low tendency to lose or gain electrons, meaning it does not readily form compounds. This is why pure gold (24-karat) does not rust, tarnish, or corrode in air or water, allowing it to retain its brilliant, yellow metallic luster for millennia. This property is the primary reason gold has been used for coinage and ornamental purposes since antiquity.
- Density and Malleability: The 79 protons contribute to a nucleus with a high positive charge, which strongly attracts the 79 surrounding electrons. This creates a powerful electrostatic force pulling the electron cloud inward, resulting in a very compact atomic structure. Combined with the metallic bonding in solid gold, this leads to an exceptionally high density (19.3 g/cm³) and remarkable malleability—a single gram can be beaten into a sheet covering nearly a square meter. These physical properties are direct outcomes of its atomic structure.
- Nuclear Applications and Medicine: While gold-197 is stable, understanding its proton count is crucial in nuclear physics. For instance, in neutron activation analysis (NAA), a sample containing gold is bombarded with neutrons. The gold-197 nucleus can absorb a neutron to become gold-198, a radioactive isotope used in certain cancer treatments (brachytherapy). The initial state—a nucleus with 79 protons—is the starting point for all such nuclear reactions and transmutations.
Scientific or Theoretical Perspective: Nuclear Stability and "Magic Numbers"
From a nuclear physics standpoint, a nucleus with 79 protons is particularly interesting. Nuclear stability is not just about the proton-to-neutron ratio; it is also influenced by the concept of "magic numbers." These are
Latest Posts
Latest Posts
-
What Is Half Of 7
Mar 21, 2026
-
Molecules Can Be Described As
Mar 21, 2026
-
Does Nucleic Acid Have Phosphorus
Mar 21, 2026
-
Number Of Protons For Gold
Mar 21, 2026
-
Lines Ab And Cg Are
Mar 21, 2026