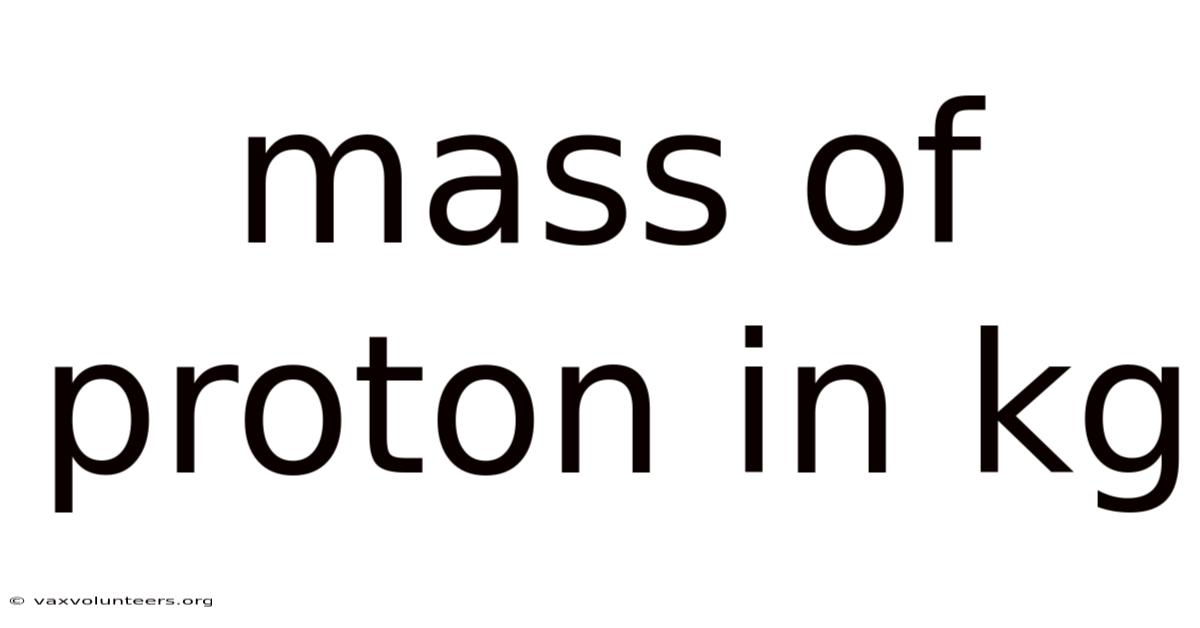Understanding the Mass of a Proton in Kilograms: A Deep Dive
At the very heart of every atom in the universe lies a tiny, positively charged sphere of matter: the proton. While it forms just one part of an atomic nucleus alongside neutrons, its properties define the very identity of chemical elements. The mass of a proton is a fundamental constant of nature, a cornerstone value upon which much of physics and chemistry is built. Yet, this seemingly simple number—approximately 1.67262192369 × 10⁻²⁷ kilograms—unlocks a profound story about the composition of matter, the forces that bind it, and the precision of modern science. This article will comprehensively explore the exact mass of a proton in kilograms, detailing not just the number itself, but its origin, how we measure it with astonishing accuracy, and why this tiny mass has colossal implications for our understanding of reality Small thing, real impact..
The proton’s mass is not merely a static figure; it is the emergent result of the dynamic dance of its internal components and the powerful forces that confine them. 67262192369(51) × 10⁻²⁷ kg**. Consider this: 007276466621 u. This level of precision—knowing the proton’s mass to better than one part in 30 billion—is a testament to decades of meticulous experimentation. In practice, the number in parentheses, (51), indicates the standard uncertainty in the last two digits, meaning the true value is expected to lie within ±0. That's why to state the CODATA 2018 recommended value is to present a pinnacle of experimental achievement: **mₚ = 1. 00000000051 × 10⁻²⁷ kg of the stated value. Which means in these units, the proton mass is approximately 1. In practice, this value is intrinsically linked to the atomic mass unit (amu or u), defined as 1/12th the mass of a carbon-12 atom. Understanding this constant is essential for calculating atomic masses, predicting nuclear reaction energies, and modeling the cosmos And that's really what it comes down to..
Worth pausing on this one.
The Detailed Explanation: What a Proton Is and Where Its Mass Comes From
To grasp the proton's mass, we must first understand what a proton is. Here's the thing — 4 MeV/c² (megaelectronvolts per speed of light squared), which is a mere 1% of the proton's total mass of approximately 938 MeV/c². The naive sum of the so-called "current quark masses" of these three quarks is only about 9.Now, it is not a fundamental particle but a composite particle, a bound state of three valence quarks: two "up" quarks and one "down" quark, held together by the strong nuclear force, mediated by particles called gluons. This glaring discrepancy is one of the most important facts in all of particle physics Most people skip this — try not to..
So, where does the other 99% of the proton's mass originate? The answer lies in quantum chromodynamics (QCD), the theory of the strong force. The interior of a proton is a seething, relativistic sea of activity. Gluons, which themselves carry the "color charge" of the strong force, are constantly being emitted and absorbed by the quarks. Adding to this, the gluon field is so intense that it spontaneously creates short-lived quark-antiquark pairs that pop in and out of existence. Plus, this chaotic, high-energy environment is governed by Einstein’s famous equation, E=mc². The vast majority of the proton's mass arises from the kinetic energy of the quarks zipping around at near-light speeds and the potential energy stored in the incredibly powerful gluon fields that bind them. In essence, the mass of everyday matter—the mass of you, the Earth, the stars—is primarily a manifestation of the energy of the strong force, not the intrinsic mass of its constituent quarks. The Higgs mechanism, which gives fundamental particles like electrons and quarks their small "current" masses, contributes only a tiny fraction to the proton's total heft Most people skip this — try not to..
Step-by-Step: The Evolution of Measuring the Proton Mass
Determining the mass of an object 10⁻¹⁵ meters wide with a precision of parts per billion is a monumental challenge. The methods have evolved from comparative to absolute measurements.
1. Early Comparative Mass Spectrometry: The first precise measurements came from mass spectrometers, which compare the mass-to-charge ratio of ions. By measuring the cyclotron frequency of a proton in a magnetic field and comparing it to a known standard (like carbon-12), scientists could determine the proton's mass relative to the atomic mass unit. This established the proton's mass as very close to 1 amu but lacked absolute precision in kilograms.
2. The Penning Trap Revolution: The breakthrough for absolute measurement came with the Penning trap. This device uses a combination of a strong homogeneous magnetic field and a precisely shaped electrostatic quadrupole field to confine a single charged particle, like a proton, in a tiny, ultra-stable orbit for months. The proton's motion in this trap has several characteristic frequencies: the **
the cyclotron frequency (due to circular motion in the magnetic field) and the axial frequency (due to oscillation along the trap's axis). On top of that, combining this with the independently known value of the elementary charge (from quantum Hall effect experiments) yields an absolute mass in kilograms. By measuring these frequencies with extraordinary precision—often by detecting tiny image currents induced in the trap electrodes—scientists can determine the proton's charge-to-mass ratio with unmatched accuracy. Modern Penning trap measurements have pushed the uncertainty below 1 part in 10¹⁰, making the proton mass one of the most precisely known fundamental constants Took long enough..
3. Modern Comparative Techniques & Atom-Based Methods: The frontier now lies in comparing the proton's mass to that of other light ions (like deuterons or helium-3) or even to the mass of an entire atom (such as carbon-12). By trapping different ions in the same Penning trap setup and measuring the ratio of their cyclotron frequencies, many systematic errors cancel, further improving precision. Additionally, advanced atom interferometry and spectroscopy of simple atoms (like hydrogen or muonic hydrogen) provide independent, complementary checks on the proton's mass and its internal structure through the Rydberg constant and other fundamental parameters.
Conclusion
The journey to understand the proton's mass reveals a profound truth: the tangible, inertial mass of the universe's building blocks is not a static property but a dynamic consequence of quantum fields and relativistic energy. The 99% of the proton's mass emerging from the kinetic and potential energy of gluons and sea quarks, as dictated by QCD, stands as a monumental validation of Einstein's equivalence and the non-Abelian gauge theory of the strong force. In practice, the painstaking evolution of measurement techniques—from comparative mass spectrometry to the exquisite precision of Penning traps—has transformed this philosophical insight into a quantitatively exact pillar of the Standard Model. This precision is not merely academic; it is essential for testing the self-consistency of QCD, probing for subtle deviations that might hint at new physics, and anchoring our understanding of how the energy of the primordial strong force condensed into the stable protons and neutrons that form the very matter of our world. In the end, the proton’s mass is more than a number—it is a direct measure of the binding energy that holds the nucleus together, and therefore, a key to understanding why the cosmos has the structure it does.