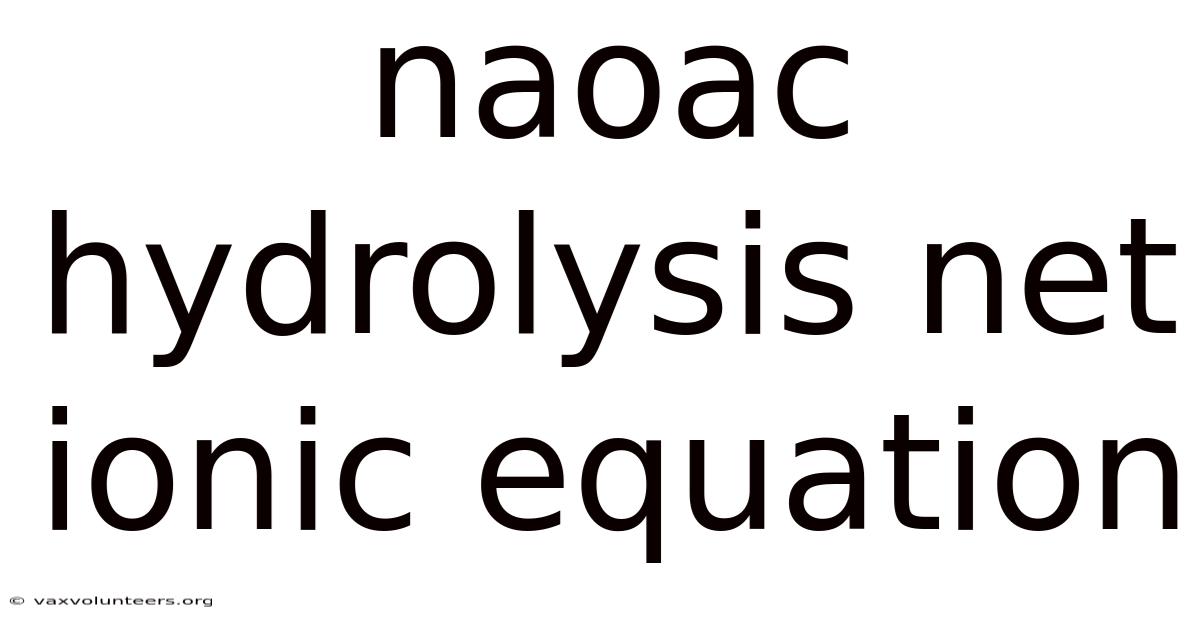Understanding the Hydrolysis of Sodium Acetate: A Complete Guide to the Net Ionic Equation
Introduction
The moment you dissolve a simple salt like sodium acetate (NaOAc) in water, the solution doesn't remain neutral. It becomes distinctly basic, a property exploited in everything from culinary chemistry to industrial buffering systems. This equation strips away the spectator ions to reveal the fundamental chemical process responsible for the increase in pH: the reaction of the acetate ion (CH₃COO⁻) with water to produce hydroxide ions (OH⁻) and acetic acid (CH₃COOH). Plus, mastering this concept is essential for predicting the pH of salt solutions, understanding buffer action, and applying equilibrium principles to real-world chemical systems. The key to understanding this behavior lies in the hydrolysis of the acetate ion, and its precise description is captured by the net ionic equation. This article will provide a thorough, step-by-step exploration of the hydrolysis of sodium acetate, culminating in a clear derivation and interpretation of its net ionic equation.
Detailed Explanation: What is Hydrolysis and Why Does NaOAc Do This?
Hydrolysis, in the context of aqueous chemistry, refers to a reaction where a substance interacts with water, causing it to split. For salts, this specifically means the reaction of an ion with water to change the pH. Whether a salt solution is acidic, basic, or neutral depends entirely on the relative strengths of the acid and base from which the salt was formed Nothing fancy..
Sodium acetate is the product of a strong base (sodium hydroxide, NaOH) and a weak acid (acetic acid, CH₃COOH). The sodium ion (Na⁺) is the conjugate acid of a strong base; it is an extremely weak acid and does not react with water to any measurable extent. This pairing is crucial. The acetate ion (CH₃COO⁻), however, is the conjugate base of a weak acid. This means acetate has a measurable affinity for protons (H⁺). It is a spectator ion. Now, when dissolved in water, some acetate ions will accept a proton from a water molecule (H₂O), a process that generates hydroxide ions (OH⁻) and leaves behind undissociated acetic acid molecules. Which means according to the fundamental principles of acid-base chemistry, the conjugate base of a weak acid is itself a weak base. This consumption of H⁺ (or production of OH⁻) is what shifts the equilibrium, resulting in a basic solution with a pH greater than 7.
Step-by-Step Breakdown: Deriving the Net Ionic Equation
Writing the net ionic equation for sodium acetate hydrolysis is a systematic process of starting with the full formula and progressively removing components that do not participate in the core reaction.
Step 1: Write the dissolution (dissociation) equation.
Solid sodium acetate is a soluble ionic compound. When it dissolves in water, it completely dissociates into its constituent ions:
NaOAc(s) → Na⁺(aq) + CH₃COO⁻(aq)
(Note: OAc is a common abbreviation for the acetate ion, CH₃COO⁻)
Step 2: Identify the ion that undergoes hydrolysis. As established, the sodium ion (Na⁺) is inert in water. The acetate ion (CH₃COO⁻) is the weak base that will react. We focus our attention on it.
Step 3: Write the molecular equation for the hydrolysis reaction.
The acetate ion reacts with water. The products are its conjugate acid (acetic acid) and the hydroxide ion:
CH₃COO⁻(aq) + H₂O(l) ⇌ CH₃COOH(aq) + OH⁻(aq)
This is a reversible reaction, denoted by the double arrow (⇌), because it reaches a chemical equilibrium. Not all acetate ions will hydrolyze; only a small fraction does Turns out it matters..
Step 4: Cancel spectator ions to form the net ionic equation.
In this specific reaction, are there any spectator ions? A spectator ion is an ion that appears unchanged on both sides of the equation. Here, the only ions explicitly shown are CH₃COO⁻ and OH⁻. The water (H₂O) is a reactant, and acetic acid (CH₃COOH) is a molecular product. There are no other ions to cancel. Because of this, the net ionic equation for the hydrolysis of sodium acetate is identical to the ionic equation from Step 3:
CH₃COO⁻(aq) + H₂O(l) ⇌ CH₃COOH(aq) + OH⁻(aq)
This simple, elegant equation is the heart of the process. It shows that the source of the basicity is the direct generation of hydroxide ions from the reaction of the acetate ion with water.
Real Examples: From Vinegar to Biological Buffers
The hydrolysis of acetate is not just a textbook exercise; it has tangible consequences.
- Household Vinegar and Baking Soda: When you mix vinegar (a dilute solution of acetic acid) with baking soda (sodium bicarbonate, NaHCO₃), you generate carbon dioxide. If you use excess vinegar, the final solution contains sodium acetate. This solution will feel slippery (a property of bases) and have a pH around 8-9, directly due to the acetate hydrolysis described by our net ionic equation.
- Acetate Buffer Systems: A classic buffer is made by mixing a weak acid (acetic acid) and its conjugate base (sodium acetate). The buffer's ability to resist pH change hinges on two simultaneous equilibria: the dissociation of acetic acid (
CH₃COOH ⇌ H⁺ + CH₃COO⁻) and the hydrolysis of acetate (our net ionic equation). The hydrolysis equilibrium constantly produces a small, steady concentration of OH⁻, which is neutralized by added acid, while the acetic acid equilibrium neutralizes added base. Understanding the hydrolysis component is key to calculating the exact pH of such a buffer using the Henderson-Hasselbalch equation. - Biological Relevance: Many biological fluids make use of acetate buffers. The pH of human blood is tightly regulated around 7.4, but other extracellular fluids or experimental culture media often employ acetate-based systems. The principles governing the hydrolysis of sodium acetate are identical to those maintaining pH stability in these sensitive environments.
Scientific or Theoretical Perspective: Equilibrium and the Kb Expression
The hydrolysis reaction is an equilibrium process governed by an equilibrium constant. For the reaction:
CH₃COO⁻(aq) + H₂O(l) ⇌ CH₃COOH(aq) + OH⁻(aq)
The base hydrolysis constant (Kb) for the acetate ion is defined as:
`Kb = [CH₃COOH][OH⁻] / [CH₃
COO⁻]Note that water is omitted from the expression because it acts as the solvent, and its activity remains effectively constant in dilute aqueous solutions. This Kb value is rarely listed in standard reference tables because it is intrinsically linked to the acid dissociation constant (Ka) of its conjugate acid, acetic acid. Through the fundamental water autoionization relationship,Ka × Kb = Kw(where Kw = 1.Now, 0 × 10⁻¹⁴ at 25°C), the base hydrolysis constant can be derived directly. Given that the Ka of acetic acid is approximately 1.Now, 8 × 10⁻⁵, the Kb for the acetate ion calculates to:Kb = Kw / Ka = (1. 0 × 10⁻¹⁴) / (1.8 × 10⁻⁵) ≈ 5.6 × 10⁻¹⁰`
This exceptionally small Kb value quantitatively confirms that acetate is a weak base, perfectly aligning with the moderately basic pH range observed in sodium acetate solutions. That said, armed with this constant, chemists can employ an ICE (Initial, Change, Equilibrium) table to solve for the equilibrium concentration of hydroxide ions, subsequently converting that value to pOH and then to pH. This mathematical framework transforms qualitative observations—like the slippery texture or litmus color change—into precise, predictable numerical outcomes.
Conclusion
The hydrolysis of sodium acetate serves as a foundational case study in aqueous acid-base chemistry, illustrating how salts derived from weak acids and strong bases inherently shift solution pH toward the basic side. That said, by tracing the process from molecular dissociation to the net ionic equation, we uncover the precise mechanism by which acetate ions extract protons from water, steadily generating hydroxide ions. Consider this: this equilibrium-driven behavior is not merely an academic curiosity; it underpins the functionality of everyday household mixtures, sophisticated laboratory buffers, and critical biological pH-regulation systems. Beyond that, the quantitative relationship between Ka, Kb, and Kw provides a reliable mathematical pathway to predict solution behavior with remarkable accuracy. The bottom line: mastering the hydrolysis of salts like sodium acetate equips students and scientists with a deeper understanding of chemical equilibrium, proving that even in seemingly simple solutions, dynamic molecular interactions continuously dictate the chemical environment. Whether designing a buffer for a sensitive enzymatic assay or simply explaining why a baking soda and vinegar reaction leaves behind a basic residue, the principles of acetate hydrolysis remain an essential and enduring tool in the chemical toolkit Still holds up..