Molecular Mass Of Mgso4 7h2o
vaxvolunteers
Feb 26, 2026 · 3 min read
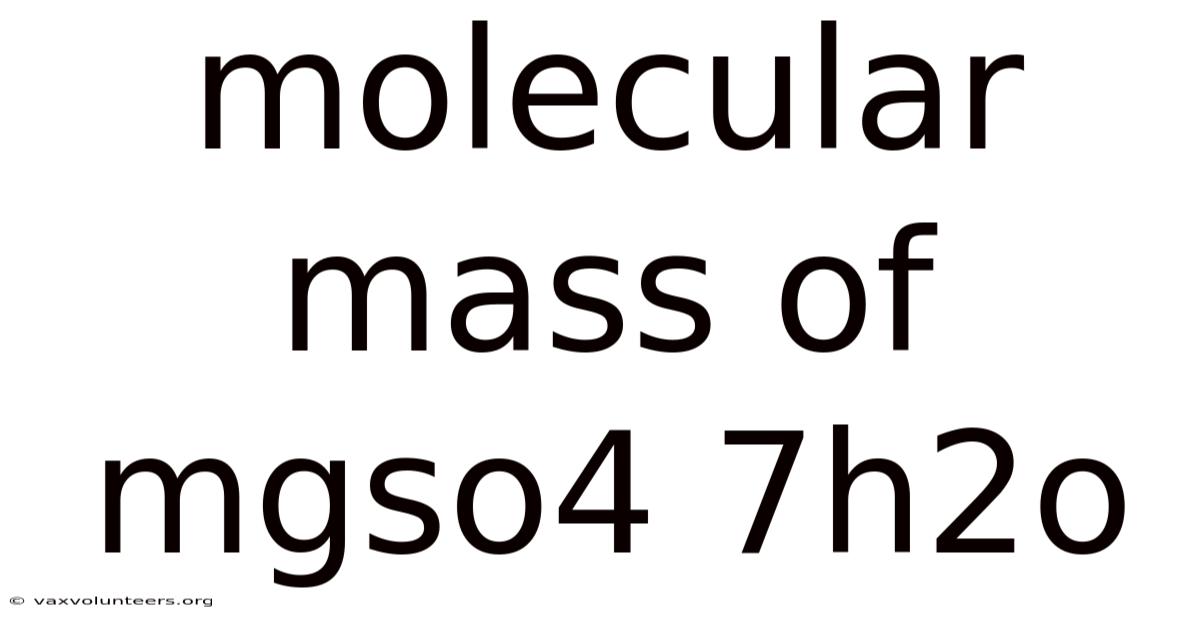
Table of Contents
Understanding the Molecular Mass of MgSO₄·7H₂O: A Comprehensive Guide
When stepping into a chemistry laboratory or studying agricultural supplements, you will inevitably encounter magnesium sulfate heptahydrate, commonly known as Epsom salt. Its chemical formula, MgSO₄·7H₂O, tells a story of a simple ionic compound intimately associated with seven molecules of water. The ability to accurately calculate its molecular mass (or more precisely for practical purposes, its molar mass) is not merely an academic exercise. It is a fundamental skill that underpins precise experimental work, industrial formulation, and even medical dosing. This article will deconstruct the concept, providing a detailed, step-by-step exploration of how to determine the molecular mass of MgSO₄·7H₂O, why this value is critical, and how to avoid common pitfalls that trip up many students and professionals alike.
Detailed Explanation: What Is MgSO₄·7H₂O and Why Does Hydration Matter?
Magnesium sulfate heptahydrate is the hydrated form of magnesium sulfate (MgSO₄). The "heptahydrate" designation means that for every one formula unit of MgSO₄, there are seven molecules of water (H₂O) crystallized within its solid structure. This is not a mere mixture; the water molecules are an integral part of the crystal lattice, held in place by hydrogen bonds and ionic interactions. In contrast, anhydrous magnesium sulfate (MgSO₄) is a hygroscopic powder that readily absorbs moisture from the air to form the heptahydrate.
The presence of these seven water molecules dramatically increases the formula mass of the compound. When we speak of the "molecular mass" of an ionic compound like this, we technically refer to its formula mass—the sum of the atomic masses of all atoms in its empirical formula. However, in common laboratory parlance, the terms "molecular mass," "formula mass," and "molar mass" are often used interchangeably for such compounds, with the understanding that the value in grams per mole (g/mol) is what we use for weighing substances. Therefore, calculating the mass of MgSO₄·7H₂O requires accounting for every single atom: one magnesium (Mg), one sulfur (S), four oxygen (O) from the sulfate ion, and an additional seven oxygen and fourteen hydrogen atoms from the seven water molecules. Ignoring the water would lead to a significant error, as the hydration shell contributes over 50% of the total mass.
Step-by-Step Breakdown: Calculating the Molecular Mass
Calculating the molar mass is a systematic process of addition, relying on standard atomic masses from the periodic table (typically using values with two decimal places for precision).
Step 1: Identify and Count All Atoms. First, dissect the formula MgSO₄·7H₂O.
- From MgSO₄: 1 Mg, 1 S, 4 O.
- From 7H₂O: 7 × (2 H + 1 O) = 14 H, 7 O.
- Total Atoms: 1 Mg, 1 S, (4+7)=11 O, 14 H.
Step 2: List Atomic Masses. Using IUPAC-accepted average atomic masses (accounting for isotopic abundance):
- Magnesium (Mg): 24.305 g/mol
- Sulfur (S): 32.06 g/mol (or 32.065)
- Oxygen (O): 15.999 g/mol (often rounded to 16.00)
- Hydrogen (H): 1.008 g/mol
Step 3: Multiply and Sum. Calculate the contribution from each element:
- Mg: 1 × 24.305 = 24.305 g/mol
- S: 1 × 32.06 = 32.06 g/mol
- O: 11 × 15.999 = 175.989 g/mol
- H: 14 × 1.008 = 14.112 g/mol
Step 4: Final Calculation. Add all contributions together: 24.305 + 32.06 = 56.365 56.365 + 175.989 = 232.354 232.354 + 14.112 = 246.466 g/mol
Rounded to two decimal places
Latest Posts
Latest Posts
-
Mm To M Scientific Notation
Feb 26, 2026
-
Which Combining Form Means Eyelid
Feb 26, 2026
-
What Is 6 120 As Percent
Feb 26, 2026
-
20 X 9 Challenge Answers
Feb 26, 2026
-
Icd 10 Foul Smelling Urine
Feb 26, 2026
Related Post
Thank you for visiting our website which covers about Molecular Mass Of Mgso4 7h2o . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.