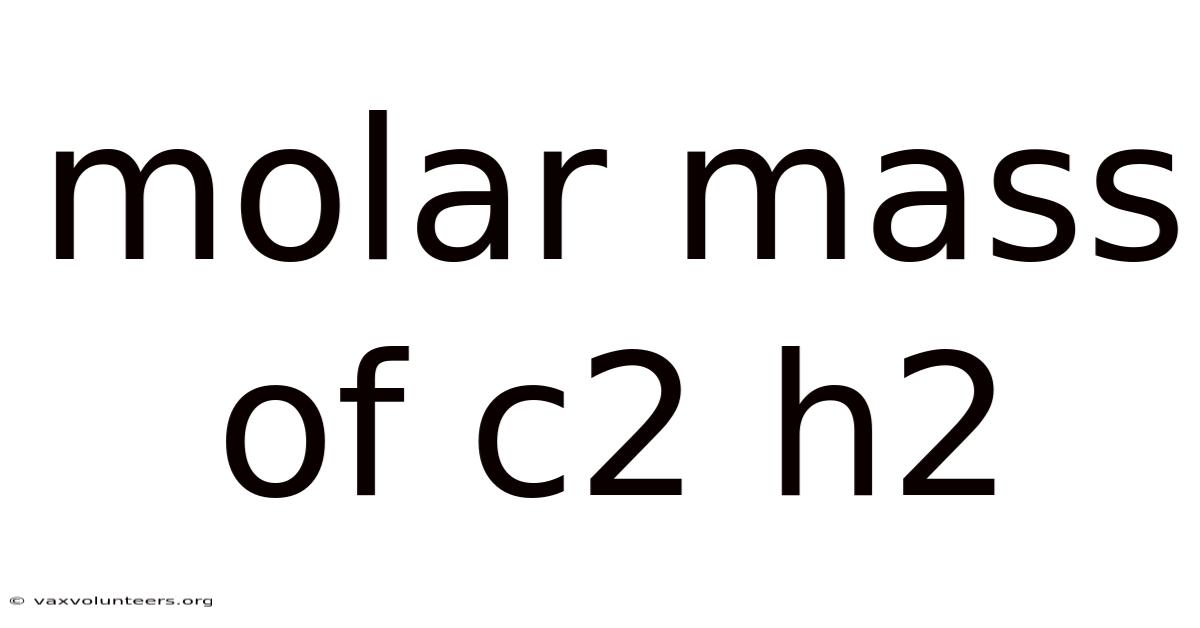Understanding the Molar Mass of C₂H₂: A Complete Guide to Acetylene's Molecular Weight
In the precise world of chemistry, where reactions hinge on exact proportions and laboratory scales measure invisible worlds, a single concept serves as a fundamental bridge between the atomic and the measurable: molar mass. For the simple yet significant molecule acetylene (C₂H₂), knowing its molar mass is not just an academic exercise; it is the key to quantifying its presence, predicting its behavior in reactions, and harnessing its powerful properties in industry. This article will provide a comprehensive, in-depth exploration of the molar mass of C₂H₂, moving from basic definitions to complex applications, ensuring you master this essential chemical calculation.
Detailed Explanation: What is Molar Mass and Why Does C₂H₂ Matter?
At its core, molar mass is the mass of one mole of a given substance, expressed in grams per mole (g/mol). Plus, 022 × 10²³). And a mole, one of the seven base units of the International System of Units (SI), is a specific number of entities—specifically, Avogadro's number (approximately 6. Because of this, the molar mass of a compound tells you exactly how many grams one "chemist's dozen" of its molecules weighs. For a molecule like acetylene (C₂H₂), this value is derived directly from the atomic masses of its constituent atoms: carbon (C) and hydrogen (H) Less friction, more output..
Acetylene, with its linear structure H-C≡C-H, is the simplest alkyne. Think about it: its importance cannot be overstated. Historically, it was the primary gas used in oxy-acetylene welding and cutting, a process that relies on the extremely high temperature (over 3,500°C) of its combustion. Today, it remains a crucial building block in organic synthesis, used to produce plastics, synthetic rubber, and various chemicals. In all these applications—whether calculating how much gas to fill a cylinder, determining reactant ratios for synthesis, or analyzing combustion products—the molar mass of C₂H₂ is the indispensable conversion factor between the microscopic world of molecules and the macroscopic world we can weigh and measure.
Step-by-Step Breakdown: Calculating the Molar Mass of C₂H₂
Calculating the molar mass of any compound follows a universal, methodical process. For C₂H₂, it involves three clear steps:
- Identify the Molecular Formula and Count Atoms: The molecular formula C₂H₂ tells us each molecule contains 2 atoms of carbon (C) and 2 atoms of hydrogen (H).
- Obtain Atomic Masses from the Periodic Table: We use the standard atomic weights, which reflect the weighted average of an element's naturally occurring isotopes.
- Atomic mass of Carbon (C): 12.011 g/mol
- Atomic mass of Hydrogen (H): 1.008 g/mol
- Multiply and Sum: Multiply the number of each type of atom by its respective atomic mass, then add the results together.
- Contribution from Carbon: 2 atoms × 12.011 g/mol = 24.022 g/mol
- Contribution from Hydrogen: 2 atoms × 1.008 g/mol = 2.016 g/mol
- Total Molar Mass of C₂H₂ = 24.022 g/mol + 2.016 g/mol = 26.038 g/mol
Because of this, one mole of acetylene molecules (6.Consider this: 022 × 10²³ molecules) has a mass of precisely 26. On the flip side, 038 grams. This value is what you will find in standard reference tables and use for all stoichiometric calculations involving acetylene Worth knowing..
Real-World Examples: Why This Number is Critical
Understanding the calculation is one thing; applying it is where true value lies. Consider these practical scenarios:
- Welding Gas Procurement: A welder needs 0.5 moles of acetylene for a large job. Using the molar mass, they can calculate the required mass: 0.5 mol × 26.038 g/mol = 13.019 grams. This ensures they purchase or fill their tank with the correct amount, avoiding project delays or hazardous over-pressurization.
- Stoichiometry in Synthesis: In a laboratory, a chemist might synthesize vinyl chloride (C₂H₃Cl) from acetylene: C₂H₂ + HCl → C₂H₃Cl. To use 10.0 grams of acetylene, they must first convert this mass to moles: 10.0 g / 26.038 g/mol ≈ 0.384 moles. This mole value then directly determines the theoretical yield of vinyl chloride, as the reaction has a 1:1 molar ratio. Without the molar mass, this quantitative planning would be impossible.
- Gas Law Applications: Acetylene is often stored under pressure in specialized cylinders. If a chemist collects 2.50 liters of acetylene gas at standard temperature and pressure (STP), they know this volume contains 2.50 L / 22.4 L/mol ≈ 0.1115 moles. The mass of this gas sample is then 0.1115 mol × 26.038 g/mol ≈ 2.90 grams. This connects gas volume measurements directly to tangible mass.
Scientific and Theoretical Perspective: Beyond the Simple Calculation
The value 26.038 g/mol is not arbitrary; it is rooted in fundamental physics and chemistry. That said, the atomic masses used (12. Now, 011 for C, 1. 008 for H) are isotopic abundances. Carbon-12 (exactly 12 u by definition) is the most abundant, but carbon-13 (13.003 u) and trace carbon-14 contribute slightly to the average. Hydrogen's mass is dominated by protium (¹H, ~1.0078 u), with small contributions from deuterium (²H or D, ~2.Now, 014 u). The molar mass is the weighted average of these isotopes as they exist in a standard terrestrial sample.
Adding to this, the concept of molar mass is intrinsically linked to Avogadro's number. Still, 022 × 10²³ mol⁻¹ ≈ 4. The mass of a single C₂H₂ molecule is its molar mass divided by Avogadro's number: 26.038 g/mol / 6.This minuscule mass is why we cannot weigh individual molecules and must use the mole concept for practical chemistry. 324 × 10⁻²³ grams. The precision of the molar mass also matters in high-precision applications like mass spectrometry, where the exact isotopic pattern of C₂H₂ (peaks at m/z 26, 27, 28) is used to identify it and confirm its structure.
Common Mistakes and Misunderstandings
Even seasoned students can stumble on molar mass calculations. Here are critical clarifications:
- Molar Mass vs. Molecular Mass: These are not the same. Molecular mass (or molecular weight) is the sum of the
Common Mistakes and Misunderstandings (Continued)
Even seasoned students can stumble on molar mass calculations. Here are critical clarifications:
- Molar Mass vs. Molecular Mass: These are not the same. Molecular mass (or molecular weight) is the sum of the atomic masses of the atoms in a single molecule of a compound. It is expressed in atomic mass units (u or amu). Here's one way to look at it: the molecular mass of acetylene (C₂H₂) is calculated as: (2 × 12.011 u) + (2 × 1.008 u) + (1 × 35.45 u) = 26.038 u. This value represents the mass of one acetylene molecule.
- Molar Mass vs. Molecular Mass: Molar mass is the mass of one mole of a substance (Avogadro's number of particles). It is expressed in grams per mole (g/mol). It is numerically equal to the molecular mass (in u) but has different units and a different meaning. The molar mass of acetylene is 26.038 g/mol. This is the mass you would measure for 6.022 × 10²³ molecules of acetylene.
The Crucial Difference: The key distinction lies in the quantity and the units. Molecular mass tells you the mass of a single molecule (26.038 u). Molar mass tells you the mass of a huge collection of molecules (26.038 g) that contains exactly 6.022 × 10²³ molecules. Confusing these concepts leads to significant errors in stoichiometric calculations and mass determinations.
The Enduring Significance of 26.038 g/mol
The value 26.So naturally, 038 g/mol for acetylene is far more than a simple number; it is a cornerstone of quantitative chemistry. Its derivation from precise atomic masses reflects the isotopic complexity of the elements involved. Its application bridges the microscopic world of atoms and molecules with the macroscopic world of grams and liters, enabling chemists to design syntheses, predict yields, manage gas storage safely, and analyze complex mixtures with remarkable accuracy. Understanding this fundamental constant, its precise determination, and the common pitfalls surrounding its use is essential for any scientist working with chemical quantities. It transforms abstract atomic scales into tangible, measurable reality, underpinning both theoretical understanding and practical industrial processes.
Short version: it depends. Long version — keep reading.
Conclusion:
The molar mass of acetylene, precisely calculated as 26.While molecular mass (26.Practically speaking, 038 g/mol, is a fundamental constant derived from the weighted average atomic masses of carbon and hydrogen, accounting for isotopic abundances. This value is indispensable across chemistry, enabling accurate stoichiometric calculations for synthesis (like determining the theoretical yield of vinyl chloride from acetylene), precise gas law applications (converting gas volumes to measurable masses), and sophisticated analytical techniques like mass spectrometry. Even so, 038 u) represents the mass of a single molecule, molar mass (26. 038 g/mol) represents the mass of one mole of molecules, connecting the microscopic and macroscopic worlds Surprisingly effective..
No fluff here — just what actually works.
Continuing from the point where the distinction between molar and molecular mass was highlighted, it is useful to examine how this numerical value translates into real‑world laboratory and industrial practices. When acetylene is employed as a feedstock for the production of vinyl chloride, the stoichiometric ratio derived from its molar mass allows engineers to calculate the exact amount of calcium carbide or methane required to generate a target volume of gas under specified temperature and pressure conditions. In gas‑phase reactors, the ideal‑gas law (PV = nRT) is routinely combined with the molar mass to convert measured flow rates (in L min⁻¹) into mass flow rates (g h⁻¹), ensuring precise control of reactant concentrations and minimizing the risk of explosive acetylene accumulation Took long enough..
Analytical chemists also rely on the 26.Isotopic subtleties further refine this process: although the standard molar mass assumes natural abundances of ^12C (≈98.On top of that, 038 g/mol value when calibrating mass spectrometers or gas chromatography detectors. In real terms, by introducing a known quantity of acetylene—often generated in situ from a calibrated solid source—the instrument’s response factor can be anchored to a traceable mass basis, which improves the accuracy of quantification for complex mixtures such as petrochemical streams or environmental samples. 9 %) and ^1H (≈99.985 %), laboratories working with enriched ^13C‑acetylene or deuterated analogues must adjust the value accordingly, underscoring the importance of recognizing that the quoted figure is an average rather than an immutable constant.
Safety considerations provide another compelling reason to master this conversion. And by converting permissible pressure limits (e. Acetylene’s propensity for spontaneous decomposition above 15 psi necessitates strict limits on the mass that can be stored in a given volume. g., 15 psi ≈ 1.03 bar) into a maximum allowable mass using the molar mass, safety officers can quickly determine whether a cylinder exceeds the hazardous threshold, thereby preventing catastrophic failures It's one of those things that adds up..
In educational settings, the acetylene example serves as a bridge between abstract atomic theory and tangible laboratory work. Students who practice converting between grams, moles, and volumes for this simple hydrocarbon develop a strong intuition for dimensional analysis—a skill that transfers without friction to more complex molecules and multi‑step syntheses.
Conclusion:
The molar mass of acetylene, 26.038 g/mol, is far more than a static number etched on a periodic table; it is a versatile tool that links the microscopic realm of atoms to the macroscopic measurements that drive synthesis, analysis, and safety. By appreciating its origins in isotopic composition, mastering its use in stoichiometric and gas‑law calculations, and recognizing the contexts where adjustments are necessary, scientists and engineers can harness acetylene’s reactivity with confidence and precision. This understanding not only prevents costly errors but also deepens the conceptual foundation upon which all quantitative chemistry rests The details matter here..