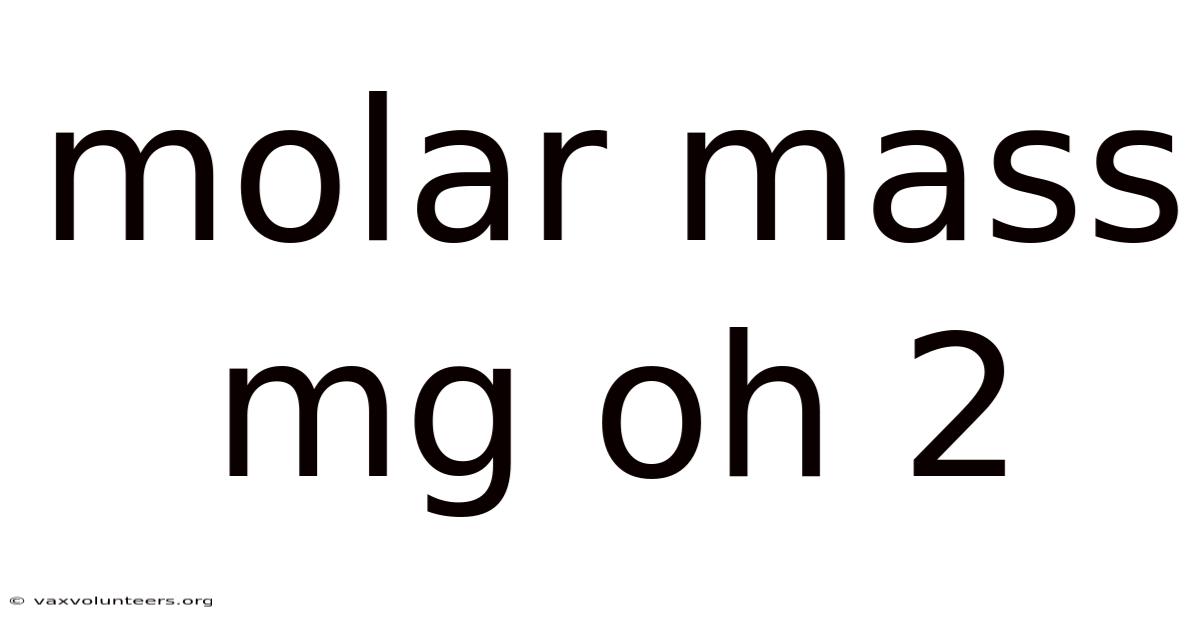Understanding the Molar Mass of Mg(OH)₂: A full breakdown
Have you ever wondered how chemists precisely measure out ingredients for a reaction or determine the correct dosage of a medication? The answer lies in a fundamental concept: molar mass. This seemingly simple calculation is the bridge between the invisible world of atoms and molecules and the tangible, measurable world of grams and liters in the laboratory. In practice, for a compound like magnesium hydroxide, Mg(OH)₂—a common antacid and wastewater treatment agent—knowing its molar mass is not just an academic exercise; it is a critical tool for accurate scientific and industrial work. This article will provide a complete, in-depth exploration of how to calculate, understand, and apply the molar mass of Mg(OH)₂, ensuring you grasp both the "how" and the "why" behind this essential chemical constant.
Detailed Explanation: What is Molar Mass and Why Does Mg(OH)₂ Matter?
At its core, the molar mass of a substance is the mass of one mole of that substance. 022 x 10²³) of entities, whether they are atoms, molecules, ions, or formula units. Also, for an ionic compound like Mg(OH)₂, which exists as a crystal lattice of Mg²⁺ and OH⁻ ions in the solid state, we refer to the mass of one mole of its formula units. The unit for molar mass is grams per mole (g/mol). A mole, in turn, is a specific number of particles—specifically, Avogadro's number (6.It is a weighted average, derived from the atomic masses of the constituent elements found on the periodic table, which themselves account for the natural isotopic abundance of each element.
Magnesium hydroxide, Mg(OH)₂, is a perfect case study. It is a white, powdery solid with low solubility in water, famously known as the active ingredient in "milk of magnesia" used as an antacid and laxative. Industrially, it
plays a vital role in flue gas desulfurization, where it neutralizes sulfur dioxide emissions from power plants, and serves as an effective flame retardant in polymers and construction materials. Across these diverse applications, precise stoichiometric calculations are mandatory, and they all begin with an accurate determination of the compound’s molar mass Worth keeping that in mind..
Step-by-Step Calculation of Mg(OH)₂ Molar Mass
Determining the molar mass of magnesium hydroxide requires breaking down its chemical formula into its constituent elements and summing their respective atomic masses. The formula Mg(OH)₂ indicates one magnesium atom, two oxygen atoms, and two hydrogen atoms. Crucially, the subscript "2" outside the parentheses applies to every element within the hydroxide group, meaning there are two OH⁻ units per formula unit Took long enough..
This is where a lot of people lose the thread.
Using standard atomic masses from the periodic table (typically rounded to two decimal places for general laboratory work):
- Magnesium (Mg): 24.On the flip side, 31 g/mol
- Oxygen (O): 16. 00 g/mol
- Hydrogen (H): 1.
The calculation proceeds by multiplying each element's atomic mass by its subscript count:
- Mass from Mg: 1 × 24.31 = 24.31 g/mol
- Mass from O: 2 × 16.00 = 32.00 g/mol
- Mass from H: 2 × 1.01 = 2.
Short version: it depends. Long version — keep reading.
Summing these contributions yields: 24.00 + 2.31 + 32.02 = **58 The details matter here..
Thus, one mole of magnesium hydroxide formula units has a mass of approximately 58.On the flip side, 33 grams. This value serves as the essential conversion factor between macroscopic mass measurements and the number of chemical entities involved in a reaction.
Real-World Applications and Stoichiometry
Knowing the molar mass of Mg(OH)₂ transforms theoretical chemistry into practical problem-solving. In pharmaceutical manufacturing, producing a standardized 400 mg dose of milk of magnesia requires converting that mass into moles to guarantee consistent active ingredient concentration. In practice, using the molar mass: 0. 400 g ÷ 58.33 g/mol ≈ 0.00686 moles of Mg(OH)₂ per dose. This precision ensures both therapeutic efficacy and patient safety Worth keeping that in mind. But it adds up..
In environmental engineering, molar mass is indispensable when designing acid-neutralization systems. Consider a wastewater stream containing 1.5 moles of hydrochloric acid (HCl) that must be treated.
The stoichiometry reveals a 1:2 mole ratio between magnesium hydroxide and hydrochloric acid. 33 g/mol) gives 43.5 moles of HCl, exactly 0.Because of that, 75 moles of Mg(OH)₂ are required. Because of that, 75 grams of magnesium hydroxide. 75 mol × 58.To neutralize 1.Multiplying by the molar mass (0.Without this conversion, operators would either waste costly reagents or leave hazardous acidity untreated, potentially violating environmental regulations Easy to understand, harder to ignore..
Not obvious, but once you see it — you'll see it everywhere.
Common Pitfalls and Best Practices
Despite its straightforward nature, molar mass calculations can introduce significant errors if approached carelessly. In real terms, the most frequent mistake involves mishandling parentheses and subscripts. For Mg(OH)₂, forgetting to distribute the external "2" to both oxygen and hydrogen yields an incorrect value of 41.32 g/mol—a discrepancy that would cascade into flawed dosing or reaction yields. Always treat polyatomic ions as grouped units when subscripts are present But it adds up..
Another critical consideration is atomic mass precision. 305, O: 15.While 24.Plus, , Mg: 24. 00, and 1.999, H: 1.g.In practice, 31, 16. Think about it: the appropriate level of precision should match the significant figures required by your final application. And 01 g/mol suffice for most educational and industrial purposes, high-precision analytical work may require more exact values (e. 008). Additionally, maintain consistent rounding throughout multi-step calculations, and only round the final answer to avoid compounding arithmetic errors.
Conclusion
The molar mass of Mg(OH)₂ is far more than a static number to memorize; it is a foundational constant that bridges atomic theory and real-world chemical practice. Mastering this calculation not only sharpens quantitative reasoning but also reinforces a core principle of chemistry: precision at the molecular scale dictates reliability at the macroscopic scale. By systematically deconstructing the formula, applying accurate atomic masses, and respecting stoichiometric relationships, scientists and engineers can precisely measure, react, and apply magnesium hydroxide across medicine, manufacturing, and environmental protection. In real terms, whether you are formulating a pharmaceutical product, treating industrial effluent, or balancing a laboratory equation, the 58. 33 g/mol benchmark for Mg(OH)₂ remains an indispensable tool in your scientific workflow Not complicated — just consistent..
Beyond manual verification, contemporary chemical practice increasingly relies on integrated software platforms and laboratory information management systems (LIMS) to automate stoichiometric tracking and cross-reference theoretical masses with real-time analytical data. Yet, software is only as reliable as the foundational knowledge of the operators who configure and interpret it. These digital safeguards significantly reduce human error, particularly in high-throughput environments such as pharmaceutical manufacturing, semiconductor cleaning, and municipal water treatment. A working grasp of molar mass derivation remains essential for troubleshooting outlier readings, validating instrument calibrations, and adapting protocols when reagent grades or supplier specifications change. As green chemistry initiatives prioritize low-toxicity, naturally sourced compounds, magnesium hydroxide continues to gain traction as a sustainable alternative to sodium hydroxide and calcium oxide. Emerging quality assurance frameworks, including automated potentiometric titration and inductively coupled plasma spectroscopy, further reinforce the necessity of precise baseline calculations. Its favorable handling profile and predictable reaction kinetics make accurate mass-to-mole conversions even more critical when scaling eco-friendly processes or navigating stringent discharge permits. When empirical results consistently align with theoretical predictions, process optimization transitions from reactive troubleshooting to proactive engineering Practical, not theoretical..
At its core, chemistry is a discipline of translation: converting symbolic representations into measurable, actionable quantities. Also, the routine calculation of a compound’s molar mass may appear elementary, but it functions as the essential checkpoint between theoretical design and practical execution. As analytical standards tighten, industrial automation expands, and sustainability mandates reshape chemical manufacturing, the ability to confidently manage between molecular formulas and macroscopic measurements will only grow in importance. Which means grounding advanced techniques in fundamental arithmetic ensures that innovation proceeds with both accuracy and accountability. The bottom line: mastering these foundational computations does more than prevent costly errors—it cultivates the quantitative discipline necessary to advance safer, more efficient, and more responsible chemical practices worldwide.