Molar Mass Cu Oh 2
vaxvolunteers
Feb 26, 2026 · 7 min read
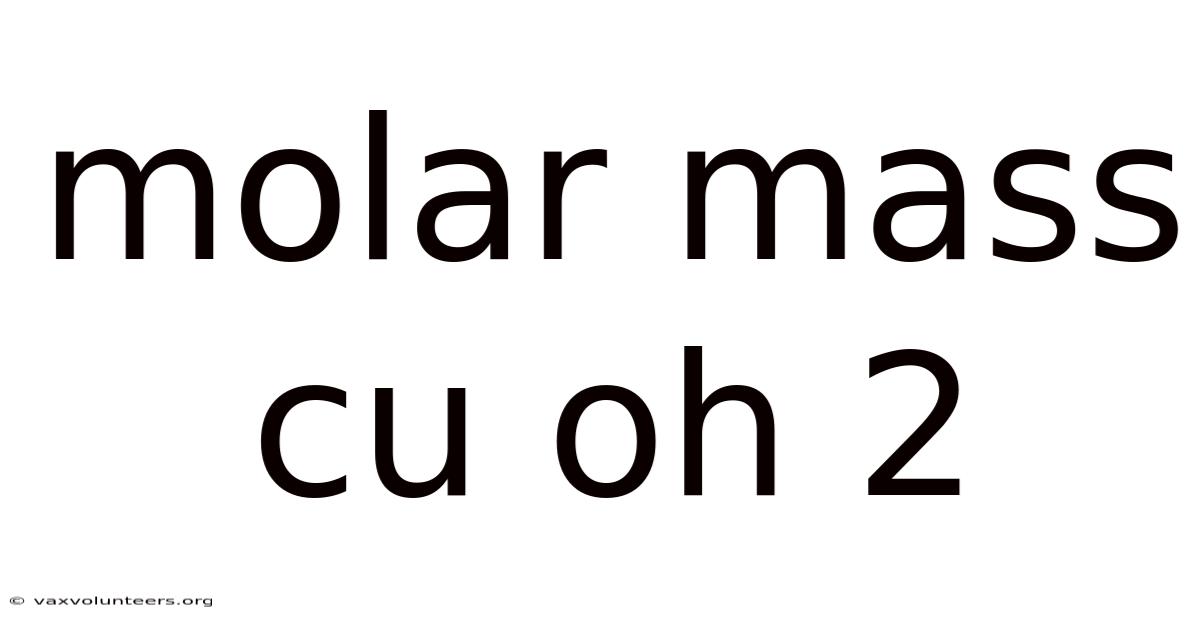
Table of Contents
Introduction
Molar mass is a fundamental concept in chemistry that represents the mass of one mole of a substance. Understanding molar mass is essential for chemical calculations, stoichiometry, and laboratory work. In this article, we will explore the molar mass of copper(II) hydroxide, Cu(OH)₂, a compound with significant applications in various chemical processes. By the end of this article, you will have a clear understanding of how to calculate its molar mass, why it matters, and how it is used in practical scenarios.
Detailed Explanation
Molar mass is defined as the mass of one mole of a substance, expressed in grams per mole (g/mol). It is calculated by summing the atomic masses of all the atoms in a molecule. For copper(II) hydroxide, Cu(OH)₂, the molar mass is determined by adding the atomic masses of copper (Cu), oxygen (O), and hydrogen (H) according to the number of atoms present in the formula.
Copper(II) hydroxide is an inorganic compound with the chemical formula Cu(OH)₂. It appears as a blue solid and is commonly used in the production of other copper compounds, as a fungicide, and in various chemical reactions. The compound consists of one copper atom, two oxygen atoms, and two hydrogen atoms. To calculate its molar mass, we need to know the atomic masses of these elements: copper (63.55 g/mol), oxygen (16.00 g/mol), and hydrogen (1.01 g/mol).
Step-by-Step Calculation of Molar Mass
To calculate the molar mass of Cu(OH)₂, follow these steps:
-
Identify the elements and their quantities in the formula:
- Copper (Cu): 1 atom
- Oxygen (O): 2 atoms
- Hydrogen (H): 2 atoms
-
Find the atomic masses of each element:
- Copper (Cu): 63.55 g/mol
- Oxygen (O): 16.00 g/mol
- Hydrogen (H): 1.01 g/mol
-
Multiply the atomic mass of each element by the number of atoms present:
- Copper: 1 × 63.55 = 63.55 g/mol
- Oxygen: 2 × 16.00 = 32.00 g/mol
- Hydrogen: 2 × 1.01 = 2.02 g/mol
-
Add the results together to get the molar mass of Cu(OH)₂:
- 63.55 + 32.00 + 2.02 = 97.57 g/mol
Therefore, the molar mass of copper(II) hydroxide, Cu(OH)₂, is 97.57 g/mol.
Real Examples and Applications
Understanding the molar mass of Cu(OH)₂ is crucial in various chemical applications. For instance, in a laboratory setting, chemists often need to prepare solutions with precise concentrations. If a chemist wants to prepare 1 liter of a 0.1 M solution of copper(II) hydroxide, they would need to calculate the mass of Cu(OH)₂ required using its molar mass.
Mass = Molarity × Volume × Molar Mass Mass = 0.1 mol/L × 1 L × 97.57 g/mol = 9.757 g
Thus, 9.757 grams of Cu(OH)₂ would be needed to prepare the solution.
In industrial applications, Cu(OH)₂ is used as a fungicide in agriculture. Knowing its molar mass allows for accurate formulation of pesticide solutions, ensuring effective and safe application rates.
Scientific and Theoretical Perspective
From a theoretical standpoint, molar mass is directly related to Avogadro's number (6.022 × 10²³), which represents the number of particles in one mole of a substance. The molar mass of Cu(OH)₂ (97.57 g/mol) means that 97.57 grams of the compound contains 6.022 × 10²³ formula units of Cu(OH)₂.
This relationship is fundamental in stoichiometry, where balanced chemical equations are used to predict the amounts of reactants and products in chemical reactions. For example, in the decomposition reaction of copper(II) hydroxide:
2 Cu(OH)₂ → CuO + H₂O + CuO
Understanding the molar mass allows chemists to calculate the exact amounts of reactants needed and products formed, ensuring efficient and safe chemical processes.
Common Mistakes and Misunderstandings
One common mistake when calculating molar mass is forgetting to account for the number of atoms of each element in the formula. For Cu(OH)₂, some might mistakenly use the atomic mass of oxygen and hydrogen without multiplying by their respective quantities (2 in this case).
Another misunderstanding is confusing molar mass with molecular mass. While both terms are related, molar mass refers to the mass of one mole of a substance (in grams per mole), whereas molecular mass is the mass of a single molecule (in atomic mass units, amu).
It's also important to use the correct atomic masses from the periodic table. Rounding errors can accumulate, so it's best to use precise values (e.g., Cu = 63.55 g/mol, not 64 g/mol) for accurate calculations.
FAQs
1. What is the molar mass of Cu(OH)₂? The molar mass of copper(II) hydroxide, Cu(OH)₂, is 97.57 g/mol. This is calculated by adding the atomic masses of one copper atom (63.55 g/mol), two oxygen atoms (2 × 16.00 g/mol), and two hydrogen atoms (2 × 1.01 g/mol).
2. Why is it important to know the molar mass of a compound? Knowing the molar mass is essential for various chemical calculations, including preparing solutions with specific concentrations, performing stoichiometric calculations in reactions, and determining the amount of substance needed for a reaction.
3. How do you calculate the molar mass of a compound? To calculate the molar mass, identify the elements in the compound and their quantities, find the atomic masses of each element from the periodic table, multiply each atomic mass by the number of atoms present, and sum the results.
4. Can molar mass be used to convert between grams and moles? Yes, molar mass serves as a conversion factor between grams and moles. The formula is: moles = mass (g) / molar mass (g/mol). This allows chemists to convert between the mass of a substance and the number of moles.
Conclusion
Understanding the molar mass of copper(II) hydroxide, Cu(OH)₂, is a fundamental aspect of chemistry that has wide-ranging applications in both academic and industrial settings. By knowing that its molar mass is 97.57 g/mol, chemists can accurately perform calculations related to solution preparation, stoichiometry, and chemical reactions. This knowledge ensures precision in experiments, efficiency in industrial processes, and a deeper understanding of chemical principles. Whether you're a student learning the basics of chemistry or a professional working in a laboratory, mastering the concept of molar mass is essential for success in the field.
Conclusion
Understanding the molar mass of copper(II) hydroxide, Cu(OH)₂, is a fundamental aspect of chemistry that has wide-ranging applications in both academic and industrial settings. By knowing that its molar mass is 97.57 g/mol, chemists can accurately perform calculations related to solution preparation, stoichiometry, and chemical reactions. This knowledge ensures precision in experiments, efficiency in industrial processes, and a deeper understanding of chemical principles. Whether you're a student learning the basics of chemistry or a professional working in a laboratory, mastering the concept of molar mass is essential for success in the field. Additionally, being mindful of common misconceptions, such as confusing molecular and molar mass or neglecting to account for the number of atoms of each element, further enhances the accuracy and reliability of chemical calculations.
Understanding the molar mass of copper(II) hydroxide, Cu(OH)₂, is a fundamental aspect of chemistry that has wide-ranging applications in both academic and industrial settings. By knowing that its molar mass is 97.57 g/mol, chemists can accurately perform calculations related to solution preparation, stoichiometry, and chemical reactions. This knowledge ensures precision in experiments, efficiency in industrial processes, and a deeper understanding of chemical principles. Whether you're a student learning the basics of chemistry or a professional working in a laboratory, mastering the concept of molar mass is essential for success in the field. Additionally, being mindful of common misconceptions, such as confusing molecular and molar mass or neglecting to account for the number of atoms of each element, further enhances the accuracy and reliability of chemical calculations.
Latest Posts
Latest Posts
-
What Is 25 Of 18
Feb 26, 2026
-
You Should Consider Your Audience
Feb 26, 2026
-
Pertaining To Above The Stomach
Feb 26, 2026
-
Which Item Completes The List
Feb 26, 2026
-
Highway Hypnosis Is Related To
Feb 26, 2026
Related Post
Thank you for visiting our website which covers about Molar Mass Cu Oh 2 . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.