Lewis Dot Structure For Sr
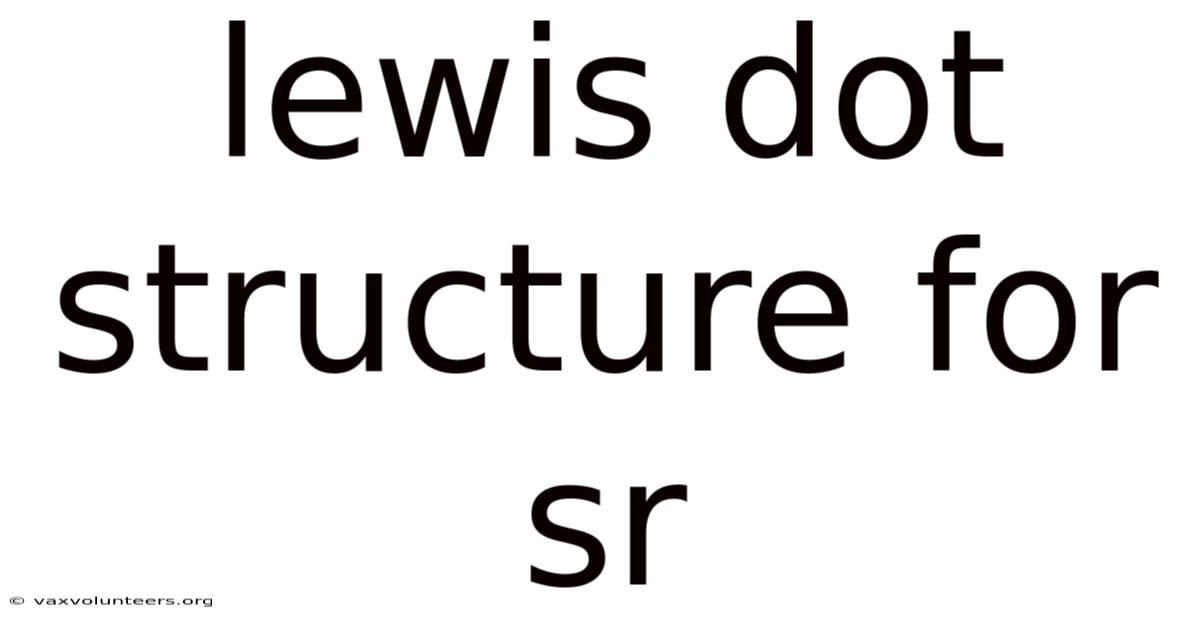
Understanding the Lewis Dot Structure for Strontium (Sr)
Introduction
In the foundational language of chemistry, the Lewis dot structure (or Lewis symbol) serves as a simple yet powerful visual shorthand. It represents an element's symbol surrounded by dots, each dot symbolizing one of the element's valence electrons—the outermost electrons involved in chemical bonding. For most main group elements, this diagram is the first step in predicting how atoms will interact to form molecules or ionic compounds. When we turn our attention to strontium (Sr), a silvery-white alkaline earth metal, its Lewis dot structure presents a beautifully straightforward case study. Strontium's position in Group 2 of the periodic table means it possesses exactly two valence electrons. Therefore, its Lewis dot structure is simply the symbol Sr with two dots placed adjacent to it. This minimalist representation is not a sign of simplicity in its chemical behavior but a direct reflection of its fundamental electronic architecture and its strong tendency to lose those two electrons to achieve a stable, noble gas electron configuration. Understanding this single, elegant diagram unlocks insights into strontium's high reactivity, its role in ionic compounds, and its place within the broader trends of the periodic table.
Detailed Explanation: Valence Electrons and the Alkaline Earth Metals
To fully grasp the Lewis dot structure for Sr, one must first understand the principles behind Lewis structures in general. Developed by Gilbert N. Lewis in the early 20th century, this system focuses on the octet rule—the observation that atoms tend to gain, lose, or share electrons to achieve a full outer shell of eight electrons (or two for hydrogen and helium), mimicking the stable configuration of the nearest noble gas. The dots in a Lewis structure represent these valence electrons, which reside in the outermost s and p orbitals. For main group elements, the group number (using the 1-18 numbering system) typically indicates the number of valence electrons. Group 1 elements have one, Group 13 have three, Group 14 have four, and so on.
Strontium, with an atomic number of 38, has an electron configuration of [Kr] 5s². The noble gas krypton (Kr) represents a stable core of 36 electrons. The two electrons in the 5s orbital are strontium's valence electrons. This places strontium firmly in Group 2, the alkaline earth metals, which include beryllium (Be), magnesium (Mg), calcium (Ca), strontium (Sr), barium (Ba), and radium (Ra). All these elements share the characteristic ns² valence electron configuration. Consequently, their Lewis dot structures are uniformly simple: the element's symbol with two dots. These dots can be placed on any side—top, bottom, left, or right—but are conventionally shown on one side for clarity. For strontium, it is written as Sr with two dots, often paired together like Sr or placed separately as Sr with one dot on the left and one on the right. This uniformity across the group highlights a key periodic trend: as you move down Group 2, the principal quantum number (n) of the valence shell increases (from n=2 for Be to n=6 for Ra), but the number of valence electrons remains constant at two.
Step-by-Step: Constructing the Lewis Dot Structure for Sr
Creating the Lewis dot structure for any element follows a logical, repeatable process. For strontium, this process is exceptionally direct due to its metallic nature and group position.
- Identify the Element and its Group: First, locate strontium (Sr) on the periodic table. It is in period 5 and Group 2. This group number is the most critical piece of information.
- Determine the Number of Valence Electrons: For main group elements, the group number equals the number of valence electrons. Since Sr is in Group 2, it has 2 valence electrons.
- Write the Symbol and Add Dots: Write the chemical symbol for the element: Sr. Then, add one dot for each valence electron. These dots are placed around the symbol. The standard convention is to place them on one side, often paired, to denote they are in the same orbital (the 5s orbital). The final structure is: Sr with two dots, typically written as Sr or Sr•• (with the dots adjacent to the 'Sr').
It is crucial to note that this process applies to the neutral atom. In chemical reactions, strontium almost invariably loses these two valence electrons to form the Sr²⁺ cation. The Lewis structure for the ion is simply the symbol Sr²⁺, with no dots, representing a stable electron configuration identical to that of krypton (Kr). This tendency to form a +2 ion is the defining chemical behavior predicted by its simple two-dot Lewis symbol.
Real Examples: Strontium in Chemical Compounds
The true value of the Lewis dot structure is revealed when predicting bonding. Strontium's two valence electrons make its fate clear: it will lose them. This is starkly different from nonmetals like oxygen or chlorine, which gain electrons to complete their octets.
...it readily donates them in ionic compounds. For instance, in strontium oxide (SrO), strontium transfers its two valence electrons to oxygen. The resulting ions, Sr²⁺ and O²⁻, are held together by strong electrostatic forces. The Lewis representation for this ionic compound is simply [Sr]²⁺ [:Ö:]²⁻, clearly showing the complete transfer of electrons and the achievement of stable, noble gas electron configurations for both ions. Similarly, in strontium chloride (SrCl₂), each chlorine atom accepts one electron from strontium, forming two Cl⁻ ions. The Lewis structure depicts Sr²⁺ surrounded by two Cl⁻ ions, often written as Sr²⁺ [∶Cl∶]⁻ [∶Cl∶]⁻.
This ionic behavior is a direct consequence of strontium's low electronegativity and its position far to the left of the periodic table. Unlike elements that share electrons to form covalent networks or molecules, alkaline earth metals like strontium have minimal tendency to share. Their large atomic radius and low ionization energy make electron loss energetically favorable, leading to the formation of crystalline ionic lattices in their solid compounds. This is why you will never find a Lewis structure for strontium showing it sharing electrons; its chemistry is dominated by the formation of the Sr²⁺ cation.
The practical implications of this are vast. The bright red color in fireworks comes from strontium ions (Sr²⁺) excited by heat. In medicine, strontium ranelate was used to treat osteoporosis because the Sr²⁺ ion can incorporate into bone mineral, mimicking calcium. In all these applications, the fundamental chemical identity of strontium is that of a two-electron donor, a trait perfectly encapsulated by its minimalist Lewis dot symbol and its inevitable transformation into the Sr²⁺ ion.
Conclusion
In summary, the Lewis dot structure for strontium is the epitome of simplicity—Sr••—directly reflecting its position as a Group 2 alkaline earth metal with exactly two valence electrons. This simple symbol is not an endpoint but a powerful predictor. It foretells strontium's exclusive role as an electron donor in chemical reactions, leading invariably to the formation of the stable Sr²⁺ cation and ionic compounds. From the step-by-step construction of its atomic symbol to its manifestation in real-world materials, the Lewis framework provides a clear and consistent model for understanding strontium's fundamental chemical behavior: a metal defined by its ready loss of two electrons to achieve a noble gas configuration.
Latest Posts
Latest Posts
-
Light Bands Are Made From
Mar 21, 2026
-
Amitotic Does Not Divide
Mar 21, 2026
-
What Is 10 Of 240
Mar 21, 2026
-
All Eucom Personnel Must Know
Mar 21, 2026
-
1 Ml How Many Units
Mar 21, 2026