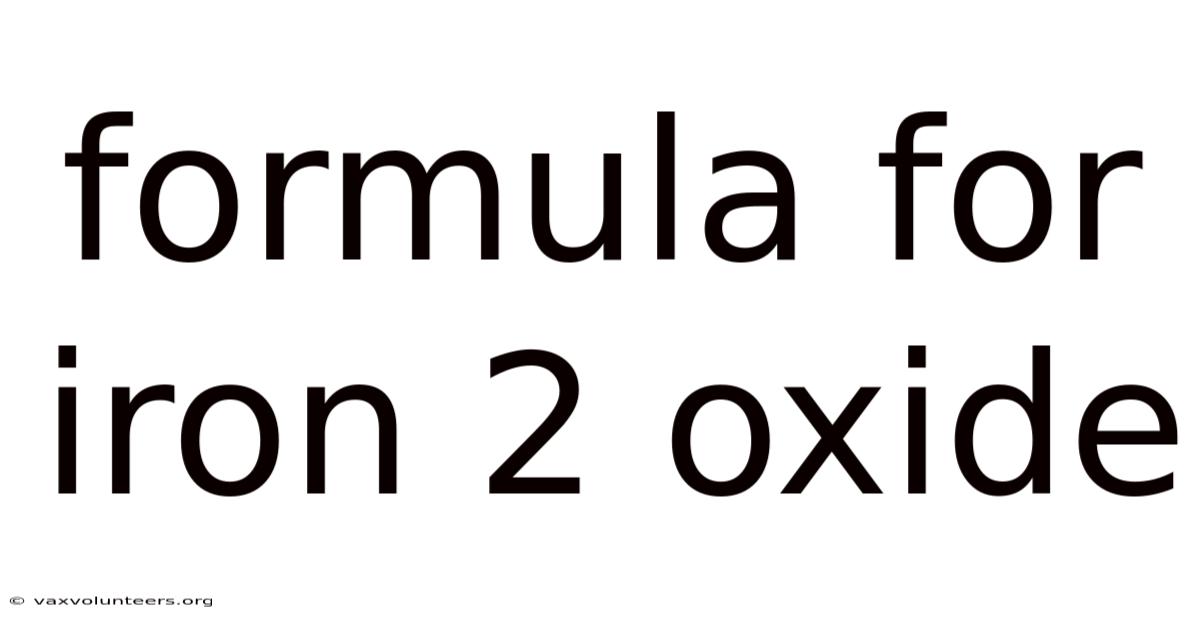The Formula for Iron(II) Oxide: More Than Just FeO
When we discuss the simple, two-word phrase "iron oxide," many of us immediately picture the familiar, flaky red-brown substance known as rust. And that common perception, however, only tells part of the story. The chemical world of iron and oxygen is rich and complex, giving rise to several distinct compounds, each with its own formula, properties, and significance. At the heart of this family lies iron(II) oxide, a compound whose deceptively simple formula, FeO, belies a fascinating story of electron transfer, crystal structure, and industrial importance. That's why understanding this formula is not merely an exercise in memorizing symbols; it is a gateway to comprehending fundamental principles of chemical bonding, oxidation states, and the very nature of inorganic materials. This article will delve deeply into the formula for iron(II) oxide, unpacking its derivation, its real-world manifestations, and why distinguishing it from its more famous cousin, iron(III) oxide, is critically important.
Detailed Explanation: Decoding FeO
To grasp the formula FeO, we must first understand the language of chemical nomenclature and the concept of oxidation states (or oxidation numbers). Even so, in iron(II) oxide, iron has an oxidation state of +2, meaning it has effectively lost two electrons. The "II" in iron(II) oxide is a Roman numeral that tells us the oxidation state of the iron atom in this compound. Oxidation state is a hypothetical charge assigned to an atom, assuming all bonds are ionic. Plus, for iron, a transition metal, this number can vary. Oxygen, in almost all its compounds (except with fluorine), has an oxidation state of -2, meaning it has gained two electrons That's the part that actually makes a difference..
The formula FeO emerges directly from the principle of electroneutrality: a stable, neutral compound must have a total charge of zero. Which means to balance this, we need an ion with a total charge of -2. The charges of the constituent ions must balance perfectly. Here's the thing — a single Fe²⁺ ion carries a +2 charge. Because of this, one iron(II) cation combines with one oxide anion to form a neutral unit: Fe²⁺ + O²⁻ → FeO. A single O²⁻ ion carries exactly a -2 charge. This 1:1 ratio is the simplest and most stable arrangement for these ions in this specific oxidation state, resulting in the empirical and molecular formula FeO Worth keeping that in mind. Simple as that..
It is here that the most critical point of confusion arises. To balance three O²⁻ ions (total charge -6), we need two Fe³⁺ ions (total charge +6), yielding the 2:3 ratio. The rust we see on old cars and bridges is primarily iron(III) oxide, often hydrated, with the formula Fe₂O₃ (or FeO(OH) for its hydrated form). In Fe₂O₃, iron has an oxidation state of +3. Day to day, **FeO is a black, powdery compound, while Fe₂O₃ is red and brittle. Which means the visual similarity of the names "iron oxide" and "iron(II) oxide" masks a profound chemical difference. ** Their magnetic properties, reactivity, and uses are entirely different, making precise terminology essential in science and industry.
Step-by-Step: Deriving the Formula
Let's walk through the logical process of determining the formula for a compound named "iron(II) oxide."
- Identify the Ions and Their Charges: The name tells us the two elements involved: iron (Fe) and oxygen (O). The "(II)" specifies the oxidation state of iron as +2, so the cation is Fe²⁺. Oxygen in oxides always has an oxidation state of -2, so the anion is O²⁻.
- Apply the Criss-Cross Method (for beginners): A common heuristic is to use the magnitude of the ion charges as subscripts for the opposite ion.
- The charge on Fe is 2. This becomes the subscript for O: O₂.
- The charge on O is 2. This becomes the subscript for Fe: Fe₂.
- This gives the preliminary formula Fe₂O₂.
- Simplify to the Lowest Whole Number Ratio: Chemical formulas are always reduced to the simplest whole-number ratio. Fe₂O₂ simplifies by dividing both subscripts by 2, yielding FeO.
- Verify Electroneutrality: Check the total charge. (1 x +2) + (1 x -2) = 0. The formula is correct and neutral.
This process highlights why the Roman numeral is non-negotiable. In real terms, without it, "iron oxide" is ambiguous and could refer to FeO, Fe₂O₃, or even mixed-valence compounds like magnetite (Fe₃O₄). The systematic name "iron(II) oxide" removes all doubt.
Real Examples and Applications
While perhaps less ubiquitous in everyday life than rust, iron(II) oxide has significant and specific applications where its unique properties are required Not complicated — just consistent. Which is the point..
- Pigments and Polishing: Finely divided FeO is used as a black pigment in some ceramics, glasses, and glazes. Its deep black color, unlike the red of Fe₂O₃, is valuable for specific aesthetic effects. It is also a component in some metal polishing compounds, where its abrasive and chemical properties aid in achieving a high shine.
- Chemical Precursor: In laboratory and industrial settings, FeO is a valuable starting material or intermediate. It can be used to synthesize other iron compounds. Here's a good example: it reacts with acids to produce iron(II) salts (like ferrous sulfate, FeSO₄), which are used in water treatment, as fertilizers, and in nutritional supplements.
- Geological and Planetary Science: FeO is a major component of wüstite, a mineral found in certain types of meteorites and in the reduced zones of some terrestrial rocks. Its presence is a key indicator of low-oxygen (reducing) conditions during formation. Studying FeO in rocks helps geologists understand the oxidation state of early Earth's mantle and the formation history of celestial bodies.
- The Thermite Reaction (A Cautionary Example): The classic thermite reaction, which produces molten iron, uses iron(III) oxide (Fe₂O₃) and aluminum powder. Still, variations exist. If one were to use FeO instead, the reaction
would proceed with a different thermodynamic profile. The reaction: 3FeO + 2Al → 3Fe + Al₂O₃ + heat is less exothermic than the standard thermite using Fe₂O₃. This is because the starting material, FeO, is already in a reduced state (Fe²⁺), so there is less driving force for the reduction and aluminum oxidation. Here's the thing — consequently, the reaction might be less violent, produce a lower temperature, and potentially yield a product iron with different purity or microstructure. This example underscores a key principle: the specific oxidation state of the metal dictates not only the name and formula but also the compound's fundamental chemical behavior and energy landscape.
This is where a lot of people lose the thread The details matter here..
This reactivity is a defining characteristic of iron(II) oxide. Unlike its more stable and inert cousin, Fe₂O₃, FeO is a potent reducing agent. It readily oxidizes in air, especially when finely divided, transforming into Fe₃O₄ or Fe₂O₃. This very instability is why pure, stoichiometric FeO is challenging to isolate and handle under ambient conditions—it "wants" to gain oxygen. Consider this: in industrial processes, this reactivity is harnessed. Here's one way to look at it: in some steelmaking slags, FeO acts as an oxygen donor, facilitating the removal of impurities like silicon and phosphorus from molten iron.
Thus, iron(II) oxide exists in a delicate balance. In real terms, it is a compound of transition, caught between the more oxidized forms of iron and the pure metal. Which means its formula, FeO, represents a simple 1:1 ratio born from clear ionic charges, yet this simplicity belies a complex identity. So it is the black pigment in a glaze, the precursor to a vital fertilizer, a mineral whisper from a reducing planetary environment, and a less-energetic variant in a fiery metal-producing reaction. Its value lies not in widespread abundance like rust, but in its specific, often niche, roles where its particular redox potential and chemical personality are precisely what is required.
Conclusion
Iron(II) oxide (FeO) serves as a perfect case study in the critical importance of precise chemical nomenclature and the profound connection between an ion's oxidation state and a compound's properties. The Roman numeral in "iron(II) oxide" is not mere formalism; it is the key that unlocks a specific formula, a predictable crystal structure, and a suite of chemical behaviors distinct from other iron oxides. While its thermodynamic instability limits its bulk use, this very reactivity makes FeO an indispensable intermediate and reagent in pigments, synthesis, geology, and specialized chemical processes. From the black depths of a ceramic glaze to the reduced zones of meteorites, FeO’s story is one of specific identity forged by the unambiguous language of oxidation states.