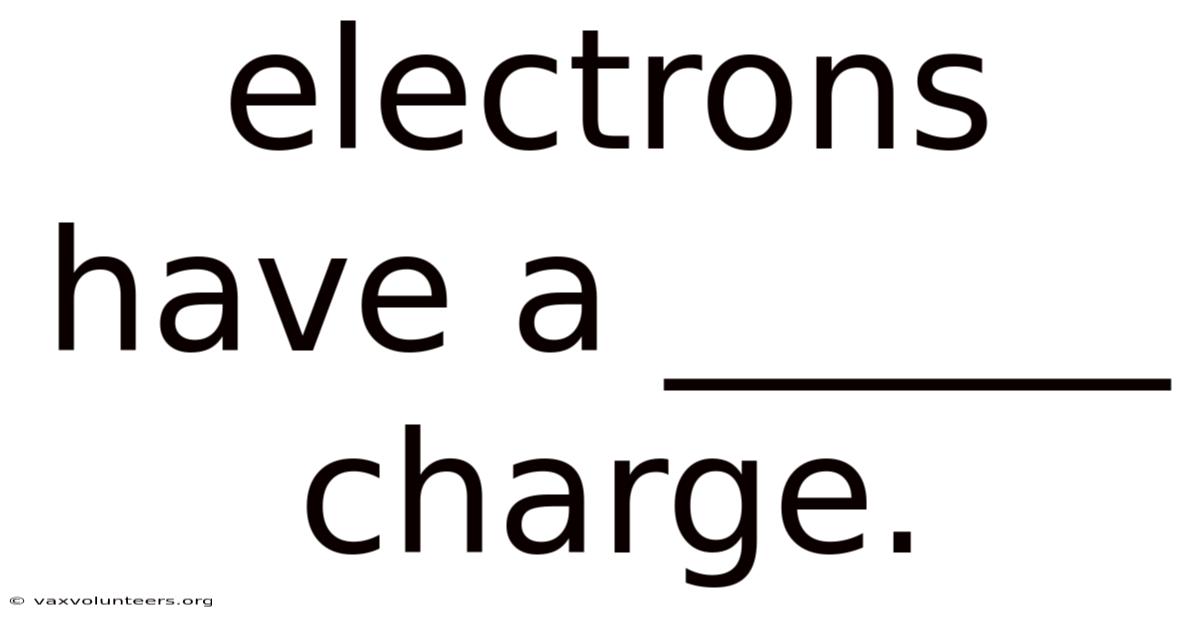Introduction: The Fundamental Property That Shapes Our Universe
At the very heart of chemistry, physics, and all of modern technology lies a deceptively simple statement: electrons have a negative charge. This foundational fact is not merely a piece of trivia; it is the keystone upon which our understanding of atomic structure, electrical currents, chemical bonding, and the behavior of matter itself is built. To grasp why electrons carry this specific type of charge is to get to the door to comprehending everything from the static cling of a balloon to the complex operations of a quantum computer. This article will delve deeply into the meaning, discovery, implications, and common misunderstandings surrounding the negative charge of the electron, providing a comprehensive view of this essential concept.
Detailed Explanation: Defining the Negative Charge
The concept of electric charge is a fundamental property of matter, much like mass. " The electron is the archetypal carrier of the negative type. Which means the convention, established by Benjamin Franklin, labels one type as "positive" and the other as "negative. 602 x 10^-19 coulombs. The magnitude of this charge is a universal constant, known as the elementary charge (e), approximately equal to 1.Basically, an electron possesses an intrinsic property that causes it to experience a repulsive force from other negatively charged objects and an attractive force toward positively charged ones. Think about it: it comes in two types: positive and negative. Every electron in the universe carries exactly this same amount of negative charge; there are no "half-charged" electrons.
This negative charge is not an add-on feature; it is inseparable from the electron's identity as a lepton, a class of fundamental particles. Now, in the standard model of particle physics, the electron's charge is a defining quantum number. It determines how the electron interacts via the electromagnetic force, one of the four fundamental forces of nature. This interaction is mediated by particles called photons. Think about it: when an electron accelerates, it emits photons, which is the basis for phenomena like radio waves and light emission from LEDs. The stability of atoms, the flow of electricity, and the very structure of molecules all depend on this precise, invariant negative charge.
Step-by-Step: The Historical Path to Discovery
Our understanding was not innate; it was the result of meticulous experimentation and theoretical leaps over centuries.
-
The Dawn of Electrostatics (1600s-1700s): Early scientists like William Gilbert and Otto von Guericke studied "static electricity" generated by rubbing materials like amber or glass. They noted two types of phenomena: objects that repelled each other and objects that attracted. Franklin's genius was in proposing a single-fluid theory: he hypothesized that an invisible fluid (charge) could be transferred between objects. He arbitrarily labeled the fluid's excess as "positive" and its deficit as "negative." This convention stuck, long before the electron was discovered.
-
Identification of the Electron (1897): J.J. Thomson's cathode ray tube experiments were central. He showed that cathode rays were composed of negatively charged particles much smaller than atoms. He measured the charge-to-mass ratio (e/m) of these "corpuscles" (later named electrons) and found it to be nearly 2000 times greater than that of a hydrogen ion, proving they were fundamental constituents of atoms. Crucially, the rays were deflected toward a positive plate, confirming the particles themselves carried a negative charge.
-
Quantifying the Charge (1909): Robert Millikan's famous oil-drop experiment provided the final, definitive proof. By balancing the gravitational force on tiny, charged oil droplets with an adjustable electric field, Millikan could measure the charge on individual droplets. He found that all charges were integer multiples of a smallest, indivisible unit: the elementary charge (e). This proved that charge is quantized and that the electron's charge was this fundamental unit, negative in sign.
Real Examples: The Negative Charge in Action
The negative charge of the electron is not an abstract idea; it is the engine of countless real-world processes Most people skip this — try not to..
- Static Electricity: When you rub a balloon on your hair, electrons are transferred from your hair to the balloon. The balloon now has an excess of electrons (a net negative charge), while your hair has a deficit (a net positive charge). The resulting attraction between the negatively charged balloon and positively charged objects (like a wall or your hand) is a direct result of the electron's charge.
- Electric Currents: In a metal wire, electrons are the mobile charge carriers. When a battery creates a voltage difference, it establishes an electric field that pushes the sea of free, negatively charged electrons in one direction. This directed flow of electrons is an electric current. The entire global power grid operates by manipulating this flow of negative charge.
- Chemical Bonding: The formation of molecules is governed by electromagnetic interactions. In a sodium chloride (NaCl) crystal, a sodium atom donates an electron to a chlorine atom. The sodium becomes a positively charged sodium ion (Na⁺), and the chlorine becomes a negatively charged chloride ion (Cl⁻). The strong electrostatic attraction between these oppositely charged ions—directly resulting from the negative charge on the gained electron—forms the ionic bond that holds the crystal together.
- Modern Electronics: Every transistor, diode, and integrated circuit functions by controlling the movement of electrons in semiconductor materials. The "N-type" semiconductor is doped with atoms that provide extra, negatively charged electrons as majority carriers. The precise manipulation of these negative charges is what allows your smartphone and computer to process information.
Scientific or Theoretical Perspective: The Quantum Framework
Classically, we can think of charge as a simple property. Quantum mechanics and quantum field theory provide a deeper, more nuanced picture Nothing fancy..
- Charge Conservation: One of the most sacred laws of physics is the conservation of electric charge. In any isolated system, the total net charge (positive minus negative) remains constant. Electrons can be created or destroyed only in particle-antiparticle pairs (an electron and a positron, its positively charged antiparticle), ensuring the net charge