Ca Oh 2 Molar Mass
vaxvolunteers
Feb 27, 2026 · 5 min read
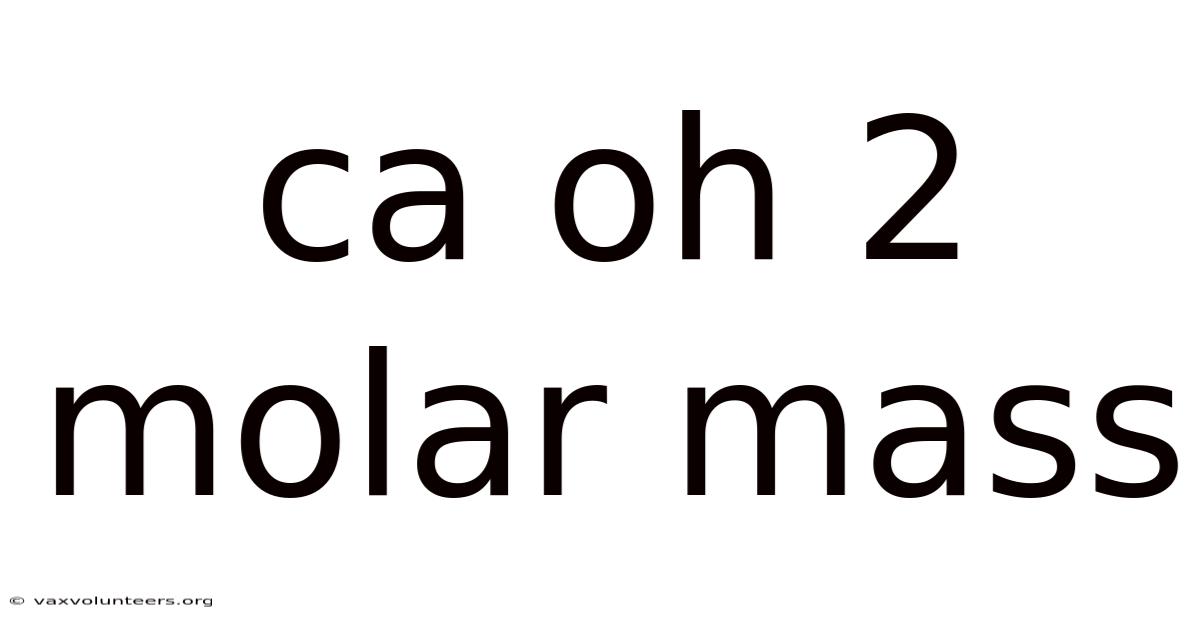
Table of Contents
Introduction
Calcium hydroxide, chemically represented as Ca(OH)₂, is a commonly used inorganic compound in various industrial, agricultural, and laboratory applications. Understanding its molar mass is essential for accurate chemical calculations, reaction stoichiometry, and formulation preparation. The molar mass of Ca(OH)₂ is the sum of the atomic masses of all atoms in one molecule of the compound. This value is critical for converting between grams and moles, a fundamental skill in chemistry. In this article, we will explore the molar mass of Ca(OH)₂ in detail, including how it is calculated, its significance, and practical applications.
Detailed Explanation
Calcium hydroxide, also known as slaked lime or hydrated lime, is formed when calcium oxide (quicklime) reacts with water. Its chemical formula, Ca(OH)₂, indicates that each molecule contains one calcium atom, two oxygen atoms, and two hydrogen atoms. The molar mass of a compound is calculated by adding the atomic masses of all the atoms present in its formula. For Ca(OH)₂, this involves summing the atomic mass of calcium (Ca), oxygen (O), and hydrogen (H). The atomic masses are based on the periodic table: calcium has an atomic mass of approximately 40.08 g/mol, oxygen is 16.00 g/mol, and hydrogen is 1.008 g/mol. By multiplying each atomic mass by the number of atoms in the formula and adding them together, we arrive at the molar mass of Ca(OH)₂.
Step-by-Step Calculation of Molar Mass
To calculate the molar mass of Ca(OH)₂, follow these steps:
- Identify the number of each type of atom in the formula: Ca(OH)₂ contains 1 calcium atom, 2 oxygen atoms, and 2 hydrogen atoms.
- Find the atomic mass of each element:
- Calcium (Ca): 40.08 g/mol
- Oxygen (O): 16.00 g/mol
- Hydrogen (H): 1.008 g/mol
- Multiply the atomic mass by the number of atoms:
- Calcium: 1 × 40.08 = 40.08 g/mol
- Oxygen: 2 × 16.00 = 32.00 g/mol
- Hydrogen: 2 × 1.008 = 2.016 g/mol
- Add the results together:
- 40.08 + 32.00 + 2.016 = 74.096 g/mol
Therefore, the molar mass of Ca(OH)₂ is approximately 74.10 g/mol when rounded to two decimal places. This value is widely used in chemical calculations and laboratory work.
Real Examples and Applications
Understanding the molar mass of Ca(OH)₂ is crucial in various real-world applications. For instance, in water treatment, calcium hydroxide is used to adjust pH levels. Accurate dosing requires knowing the molar mass to convert between the mass of the compound and the number of moles needed. In agriculture, Ca(OH)₂ is used to neutralize acidic soils. Farmers and agronomists must calculate the exact amount of lime required based on the molar mass to ensure effective soil treatment. In construction, calcium hydroxide is a key component in mortar and plaster. The molar mass helps in determining the correct proportions of materials to achieve desired properties. Additionally, in laboratory settings, Ca(OH)₂ is used in titrations and other analytical procedures, where precise molar calculations are essential for accurate results.
Scientific and Theoretical Perspective
From a theoretical standpoint, the molar mass of a compound like Ca(OH)₂ is rooted in the concept of the mole, a fundamental unit in chemistry. One mole of any substance contains Avogadro's number (6.022 × 10²³) of particles, whether atoms, molecules, or ions. The molar mass, expressed in grams per mole, allows chemists to bridge the gap between the microscopic world of atoms and the macroscopic world of measurable quantities. For Ca(OH)₂, knowing its molar mass enables the conversion of mass to moles and vice versa, facilitating stoichiometric calculations in chemical reactions. This is particularly important in reactions involving calcium hydroxide, such as its reaction with acids to form calcium salts and water. The molar mass also plays a role in determining the concentration of solutions, which is vital in both research and industrial processes.
Common Mistakes or Misunderstandings
One common mistake when calculating the molar mass of Ca(OH)₂ is forgetting to account for the subscript outside the parentheses. For example, some might incorrectly calculate the molar mass by considering only one oxygen and one hydrogen atom, rather than two of each. Another misunderstanding is confusing the molar mass with the molecular mass, which is essentially the same but expressed in atomic mass units (amu) rather than grams per mole. Additionally, using outdated or incorrect atomic masses from memory can lead to errors. It's important to always refer to the periodic table for the most accurate atomic masses. Lastly, rounding errors can accumulate if intermediate steps are rounded too early; it's best to carry extra significant figures through the calculation and round only the final answer.
FAQs
1. What is the molar mass of Ca(OH)₂? The molar mass of Ca(OH)₂ is approximately 74.10 g/mol. This is calculated by adding the atomic masses of one calcium atom (40.08 g/mol), two oxygen atoms (32.00 g/mol), and two hydrogen atoms (2.016 g/mol).
2. Why is the molar mass of Ca(OH)₂ important? The molar mass is crucial for converting between grams and moles, which is essential for chemical calculations, reaction stoichiometry, and preparing solutions with precise concentrations.
3. How do you calculate the molar mass of Ca(OH)₂? To calculate the molar mass, multiply the atomic mass of each element by the number of atoms in the formula and add the results. For Ca(OH)₂, this means: (1 × 40.08) + (2 × 16.00) + (2 × 1.008) = 74.096 g/mol.
4. Can the molar mass of Ca(OH)₂ vary? The molar mass is based on the standard atomic masses of the elements, which are relatively constant. However, isotopic variations can cause slight differences, but these are negligible for most practical purposes.
Conclusion
Understanding the molar mass of Ca(OH)₂ is a fundamental aspect of chemistry that has wide-ranging applications in industry, agriculture, and laboratory work. By accurately calculating and applying this value, chemists and professionals can ensure precise measurements, effective formulations, and successful chemical reactions. Whether you're adjusting soil pH, treating water, or conducting a titration, the molar mass of calcium hydroxide is a key piece of information that bridges the gap between theory and practice. Mastery of this concept not only enhances your chemical knowledge but also empowers you to tackle real-world challenges with confidence and accuracy.
Latest Posts
Latest Posts
-
Where A Story Takes Place
Feb 27, 2026
-
What Is 20 Of 300
Feb 27, 2026
-
Half Of 1 1 2
Feb 27, 2026
-
How Tall Is 68 Inches
Feb 27, 2026
-
Division Of 144 By 12
Feb 27, 2026
Related Post
Thank you for visiting our website which covers about Ca Oh 2 Molar Mass . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.