Air Is A ___________-____________ Solution.
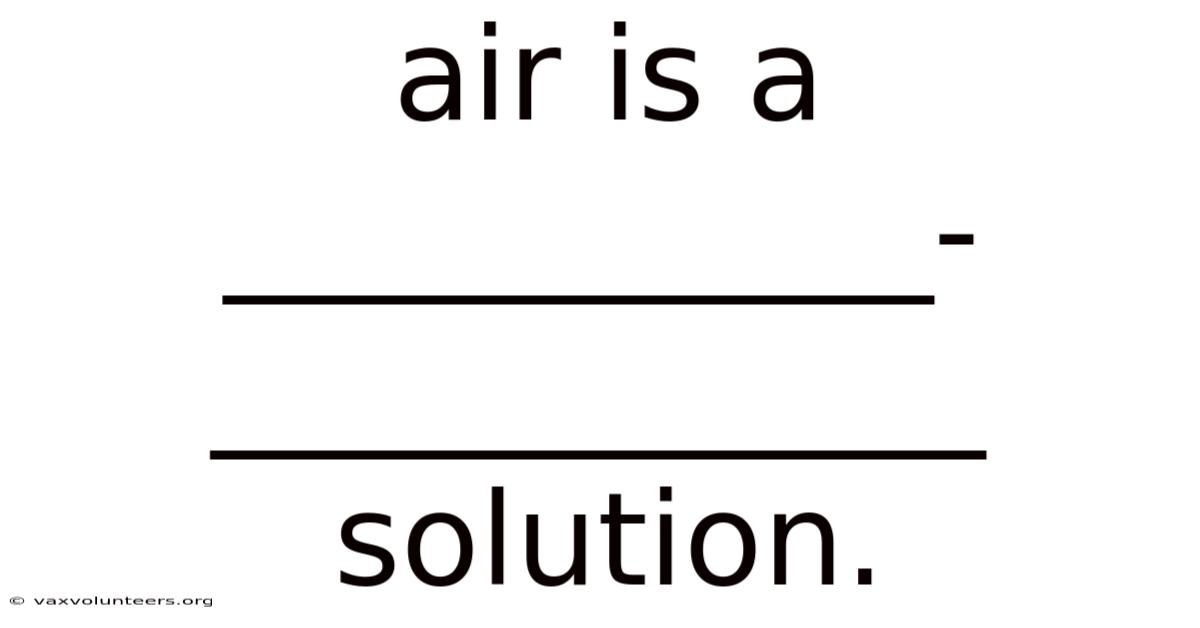
Air is a Homogeneous Solution: Understanding the Nature of Earth’s Atmosphere
Air is a homogeneous solution of gases, a fundamental concept in chemistry and environmental science. This statement, often phrased as “air is a - solution,” is a foundational idea that underpins our understanding of the atmosphere’s composition, behavior, and impact on life. While the phrase may seem simple, it encapsulates a complex interplay of scientific principles, from the properties of gases to the dynamics of Earth’s climate. In this article, we will explore what it means for air to be a homogeneous solution, delve into its components, and examine why this understanding is critical for both scientific and practical applications.
What is a Homogeneous Solution?
A homogeneous solution is a uniform mixture of two or more substances where the components are evenly distributed at the molecular level. Unlike heterogeneous mixtures, which have visibly distinct parts (like sand in water), homogeneous solutions appear as a single phase. For example, saltwater is a homogeneous solution because the salt dissolves completely into the water, creating a consistent composition throughout.
In the case of air, the term “solution” is used in a broader sense. While air is not a liquid solution, it is a gas mixture that behaves similarly to a homogeneous solution. The gases in air—primarily nitrogen, oxygen, argon, and carbon dioxide—are uniformly mixed, creating a consistent composition throughout the atmosphere. This uniformity is why air appears colorless, odorless, and transparent, even though it contains multiple substances.
The concept of a homogeneous solution is rooted in the principles of solution chemistry, which studies how solutes dissolve in solvents. In air, the “solvent” is not a liquid but the gaseous medium itself. The “solute” refers to the individual gas molecules that are dispersed throughout the atmosphere. This analogy helps explain why air is often described as a solution, even though it is not a liquid.
The Components of Air: A Complex Mixture
Air is not a single substance but a mixture of gases that vary in concentration. The primary components of dry air include:
- Nitrogen (N₂): Approximately 78% of the atmosphere, nitrogen is the most abundant gas. It is relatively inert, meaning it does not readily react with other substances.
- Oxygen (O₂): About 21% of the atmosphere, oxygen is essential for respiration in most living organisms.
- Argon (Ar): Around 0.93%, argon is a noble gas that is chemically unreactive.
- Carbon Dioxide (CO₂): Approximately 0.04%, CO₂ plays a critical role in the greenhouse effect and is a byproduct of respiration and combustion.
- Other trace gases: These include neon, helium, methane, and water vapor, which vary in concentration depending on location and environmental conditions.
The homogeneous nature of air arises from the fact that these gases are evenly distributed throughout the atmosphere. Unlike a heterogeneous mixture, where components can be separated by physical means (e.g., filtering sand from water), the gases in air cannot be easily separated without specialized techniques like fractional distillation. This uniformity is a hallmark of a homogeneous solution.
Why Is Air Considered a Homogeneous Solution?
The classification of air as a homogeneous solution is based on several key characteristics:
-
Uniform Composition: The gases in air are mixed so thoroughly that their proportions remain constant throughout the atmosphere. For example, the percentage of oxygen in the air at sea level is roughly the same as at high altitudes, though pressure and temperature variations can affect gas behavior.
-
Molecular-Level Mixing: The molecules of different gases in air are intermingled at the atomic level, creating a single phase. This is why air does not settle or separate
Beyond this molecular intermixing, the homogeneity of air is maintained by constant atmospheric motion. Winds, convection currents, and diffusion perpetually stir the gaseous mixture, preventing any significant stratification or settling of components under normal conditions. This dynamic equilibrium ensures that, on a macroscopic scale, air behaves as a single, uniform substance. However, this ideal homogeneity is a local and conditional property. Over vast vertical distances—such as from the troposphere to the stratosphere—concentrations of certain gases like ozone and water vapor change dramatically, creating distinct atmospheric layers. Similarly, on a very local scale, plumes of smoke, volcanic emissions, or even the breath we exhale create temporary, heterogeneous pockets within the otherwise uniform mixture. These exceptions are critical, as they highlight that air’s classification as a homogeneous solution applies to its bulk, well-mixed state in the lower atmosphere, not under all possible circumstances.
This perspective is not merely academic. Recognizing air as a homogeneous solution is fundamental to fields like meteorology, environmental science, and respiratory physiology. It explains why combustion engines and living lungs can operate predictably anywhere on Earth’s surface; the reactant (oxygen) is consistently available at a known partial pressure. It also underpins our understanding of how pollutants disperse and how trace gases, though minute in concentration, can exert global effects precisely because they are mixed so uniformly throughout the entire atmospheric column.
In conclusion, air is best understood as a dynamic, gaseous homogeneous solution. Its primary components—nitrogen, oxygen, and argon—are so thoroughly and continually mixed that they present a single phase with a consistent composition at any given altitude and location under stable conditions. While localized heterogeneities and vertical stratification exist, the overarching uniformity of the atmosphere is what allows it to function as the essential, life-supporting envelope of our planet. This homogeneity facilitates predictable gas exchange, climate systems, and chemical processes, making it a cornerstone concept for both natural sciences and our daily experience of the world.
The implications of this understanding are far-reaching, influencing not only our comprehension of atmospheric phenomena but also informing strategies for environmental conservation and human health. By acknowledging the homogeneous nature of air, we can better appreciate the interconnectedness of our planet's systems and the delicate balance that sustains life. Furthermore, recognizing the dynamic and conditional aspects of air's homogeneity encourages a nuanced approach to addressing global challenges, such as climate change and air pollution, which require a deep understanding of the complex interactions within our atmosphere. Ultimately, the concept of air as a homogeneous solution serves as a powerful reminder of the intricate beauty and functionality of our planet's atmospheric system, underscoring the importance of continued scientific inquiry and stewardship to preserve the integrity of this vital component of our ecosystem.
Continuing seamlessly from the provided text:
The profound implications of this understanding extend far beyond theoretical frameworks. Recognizing air's fundamental homogeneity is not merely an academic exercise; it is the bedrock upon which practical solutions to global challenges are built. In the realm of environmental conservation, this knowledge is indispensable. It allows scientists to model pollutant transport with greater accuracy, predicting how emissions from a single source might disperse across continents or oceans. This predictive capability is crucial for designing effective air quality regulations, international agreements on greenhouse gases, and strategies for mitigating the spread of transboundary pollution. Understanding the uniform mixing also highlights the global nature of climate change; trace greenhouse gases, despite their minuscule local concentrations, exert their warming influence uniformly because they are thoroughly mixed throughout the entire atmospheric column.
Similarly, in the domain of human health, the consistent availability of oxygen at predictable partial pressures is vital for the design and operation of life-support systems, from medical ventilators to high-altitude aircraft cabins. However, this very homogeneity also means that pollutants introduced into the air can be carried far from their source, affecting populations distant from the original emission point. Recognizing this interconnectedness underscores the critical importance of clean air policies and the need for global cooperation in tackling air pollution, which remains a leading environmental health risk worldwide.
Furthermore, this perspective fosters a deeper appreciation for the interconnectedness of Earth's systems. The atmosphere, hydrosphere, lithosphere, and biosphere are not isolated compartments; they are dynamically linked through processes governed, in part, by the properties of air. The uniform mixing of gases influences ocean-atmosphere exchange, nutrient cycling, and even the distribution of aerosols that seed clouds. Disrupting this delicate balance, whether through increased greenhouse gas concentrations or localized pollution hotspots, can have cascading effects throughout the entire planetary system.
Therefore, the concept of air as a dynamic, gaseous homogeneous solution is far more than a definition; it is a fundamental principle that illuminates the workings of our planet and our place within it. It empowers us to understand natural phenomena, predict environmental changes, safeguard human health, and develop strategies for sustainable stewardship. As we confront the complex challenges of the 21st century – climate change, biodiversity loss, and resource depletion – a nuanced understanding of the atmosphere's inherent properties, including its remarkable homogeneity, becomes increasingly essential. Continued scientific inquiry into the dynamics of atmospheric mixing, coupled with responsible action informed by this knowledge, is paramount to preserving the integrity of this vital, life-sustaining envelope that surrounds our planet.
Latest Posts
Latest Posts
-
Molecular Orbital Diagram For H2
Mar 26, 2026
-
What Is 10 Of 800
Mar 26, 2026
-
What Is The Real Time
Mar 26, 2026
-
What Is Called Deer Meat
Mar 26, 2026
-
6 Piece Chicken Mcnuggets Calories
Mar 26, 2026