3r 4s 3 4 Dimethylhexane
vaxvolunteers
Mar 17, 2026 · 5 min read
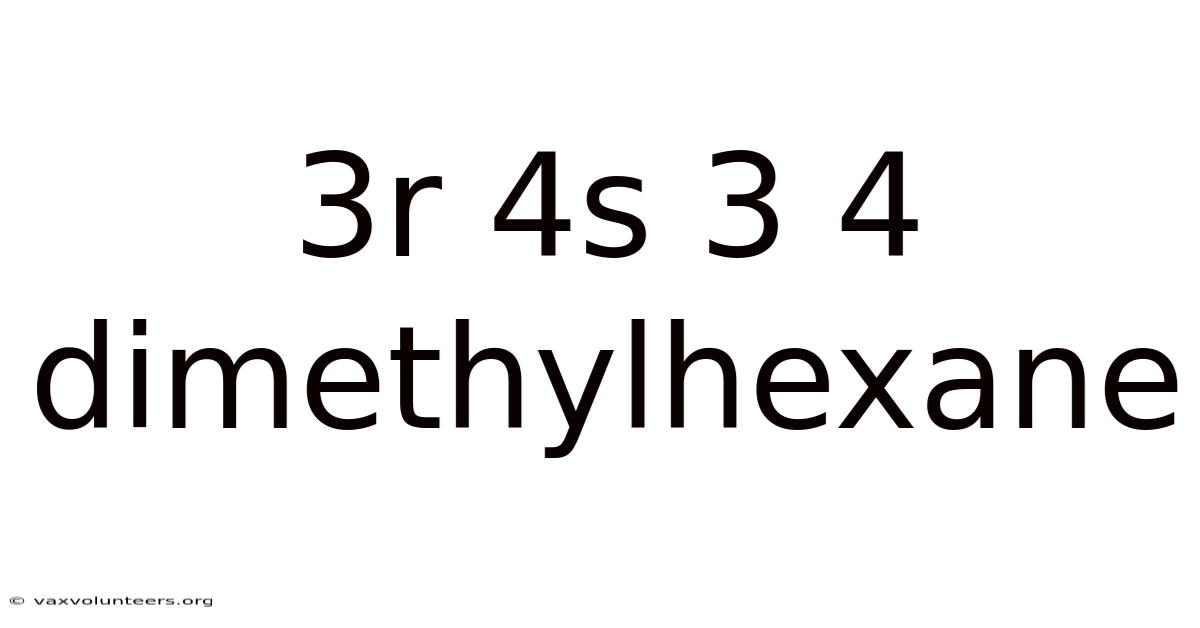
Table of Contents
Understanding 3R,4S-3,4-Dimethylhexane: A Deep Dive into Stereochemistry and Nomenclature
Introduction
In the intricate world of organic chemistry, the name of a compound is far more than a simple label; it is a precise blueprint detailing its atomic architecture and, crucially, its three-dimensional shape. The term 3R,4S-3,4-dimethylhexane exemplifies this perfectly. At first glance, it describes a branched alkane—hexane with two methyl groups attached to carbons 3 and 4. However, the prefixes "3R" and "4S" transform this from a simple structural formula into a specification of absolute configuration, revealing that this molecule exists as a single, non-superimposable mirror image at two distinct chiral centers. This article will comprehensively unpack this designation, exploring the foundational principles of stereochemistry that make such a name necessary, the step-by-step logic behind assigning R and S labels, and the profound significance this three-dimensional detail holds in scientific and industrial applications. Understanding this compound is a masterclass in how modern chemical nomenclature communicates spatial information with unambiguous precision.
Detailed Explanation: From Simple Chain to Chiral Molecule
Let us begin by dissecting the name into its core components. The parent chain is hexane, a straight-chain alkane with six carbon atoms (C6H14). The substituents are two methyl groups (-CH3), attached to the third and fourth carbon atoms of this chain. This gives us the basic structural isomer, 3,4-dimethylhexane. If we drew its simplest Lewis structure on a flat page, we might see a zigzagging carbon backbone with methyl groups projecting up or down from carbons 3 and 4. However, this flat drawing inherently obscures a critical feature: the tetrahedral geometry around each of those two internal carbons.
Carbon atoms forming four single bonds adopt a tetrahedral geometry. For a carbon atom to be a chiral center (or stereocenter), it must be bonded to four different substituent groups. In 3,4-dimethylhexane, we must examine carbons 3 and 4. Carbon 3 is bonded to: 1) a hydrogen atom, 2) a methyl group (the branch), 3) a chain fragment going toward carbon 2 (which is -CH2-CH3, an ethyl group), and 4) a chain fragment going toward carbon 4 (which is -CH(CH3)-CH2-CH3, a complex but unique group). Because all four attachments are different, carbon 3 is a chiral center. Similarly, carbon 4 is bonded to: 1) a hydrogen atom, 2) a methyl group (the branch), 3) a chain fragment toward carbon 3 (which is -CH(CH3)-CH2-CH3), and 4) a chain fragment toward carbon 5 (which is -CH2-CH3, an ethyl group). Again, all four are distinct, making carbon 4 a second chiral center.
A molecule with n chiral centers can theoretically have up to 2^n stereoisomers. Here, with two chiral centers, we might expect four possible combinations: (R,R), (R,S), (S,R), and (S,S). However, the molecule also possesses a plane of symmetry in its meso form. The (R,S) and (S,R) designations actually refer to the same meso compound because the molecule has an internal plane of symmetry that makes it achiral overall (it is superimposable on its mirror image). The (R,R) and (S,S) forms are a pair of enantiomers (non-superimposable mirror images). Therefore, 3,4-dimethylhexane exists as three distinct stereoisomers: one meso form and one pair of enantiomers. The specific name 3R,4S-3,4-dimethylhexane unambiguously identifies the meso isomer, specifying the absolute configuration at each chiral center.
Step-by-Step Breakdown: Assigning R and S Configuration
Assigning the R (Rectus, meaning "right") or S (Sinister, meaning "left") label is a systematic process governed by the Cahn-Ingold-Prelog (CIP) priority rules. Here is the logical flow for determining the configuration at carbon 3 in 3,4-dimethylhexane:
- Identify the Chiral Center: Focus on carbon atom number 3.
- List the Four Attached Groups: For C3, they are: -H, -CH3 (the branch), -CH2-CH3 (the ethyl group toward C2), and -CH(CH3)-CH2-CH3 (the complex chain toward C4).
- Assign Priorities (1 = highest, 4 = lowest): Compare the atoms directly attached to the chiral center.
- The -H atom has the lowest atomic number (1), so it gets Priority 4.
- The -CH3 group's first atom is carbon (atomic number 6).
- The -CH2-CH3 group's first atom is also carbon.
- The -CH(CH3)-CH2-CH3 group's first atom is also carbon. We have a tie. We must move outward, looking at the atoms attached to these first atoms.
- For the -CH3 branch: the carbon is bonded to three H's (atomic number 1,1,1).
- For the -CH2-CH3 (ethyl): the carbon is bonded to two H's and one C (from the next CH3). The list is (6,1,1).
- For the -CH(CH3)-CH2-CH3: the carbon is bonded to one H, one CH3, and one CH2- (from the rest of the chain). The list is (6,6,1).
Comparing these lists at the first point of difference: (6,6,1) > (6,1,1) > (1,1,1). Therefore:
- Priority 1: -CH(CH3)-CH2-CH3 (the complex chain toward C4)
- Priority 2: -CH2-CH3 (the ethyl group toward C2)
- Priority 3: -CH3 (the methyl branch)
- Priority 4: -H
- Orient the Molecule: Rotate the molecule so that the group with Priority 4 (the -H) is pointing away from you, into the plane of the page/screen.
- Determine the Direction: Observe the sequence of priorities 1 → 2 → 3.
- If this sequence runs clockwise, the configuration is R.
If the sequence runs counterclockwise, the configuration is S.
For the (R) configuration at C3, the sequence 1→2→3 must be clockwise when H is pointing away. For the (S) configuration, the sequence 1→2→3 must be counterclockwise when H is pointing away.
This same process is repeated for carbon 4 to fully specify the stereochemistry of the molecule as (3R,4S) or (3S,4R) for the meso form, or (3R,4R) and (3S,4S) for the pair of enantiomers.
Conclusion
The designation 3R,4S-3,4-dimethylhexane is a precise, unambiguous name for one specific stereoisomer of 3,4-dimethylhexane. It identifies the molecule as the meso form, where the two chiral centers have opposite configurations. This systematic nomenclature, based on the Cahn-Ingold-Prelog rules, is essential for clearly communicating the three-dimensional structure of chiral molecules in chemistry. It allows chemists to distinguish between molecules that may have the same molecular formula and connectivity but differ in the spatial arrangement of their atoms, which can have profound effects on their chemical and biological properties.
Latest Posts
Latest Posts
-
Nature Is To Nurture As
Mar 17, 2026
-
Which Best Describes Traditional Classification
Mar 17, 2026
-
Simplify The Following Polynomial Expression
Mar 17, 2026
-
How Many Ml In Ul
Mar 17, 2026
-
Terry Made A Plastic Birdhouse
Mar 17, 2026
Related Post
Thank you for visiting our website which covers about 3r 4s 3 4 Dimethylhexane . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.