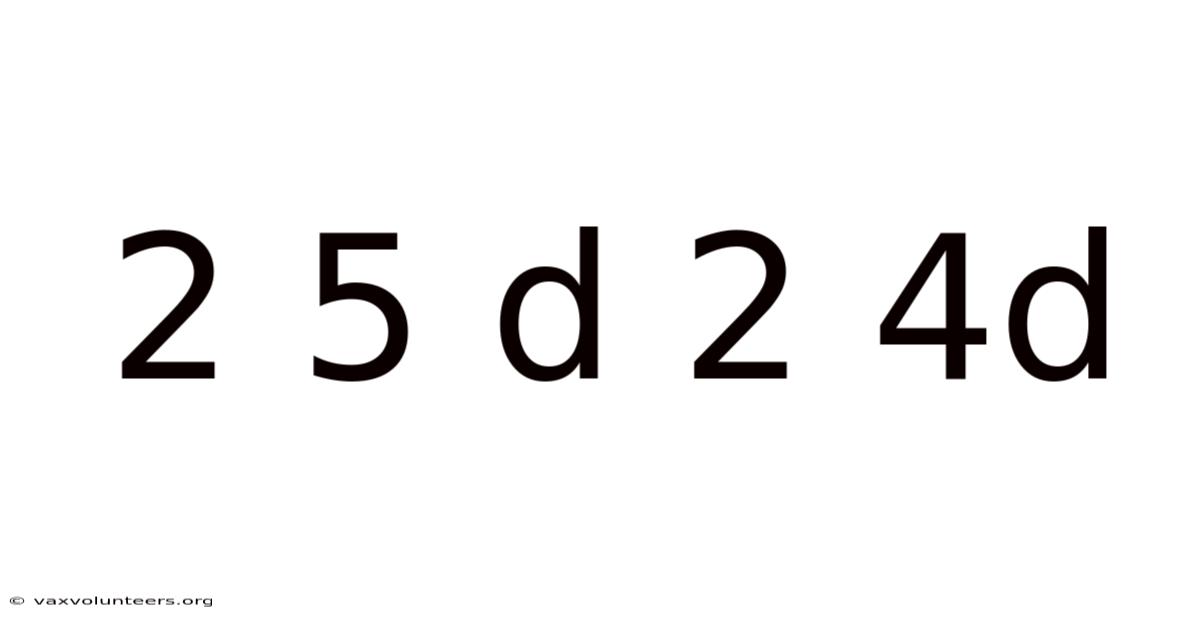Understanding Orbital Hybridization: Comparing sp³d² (3d) vs. sp³d² (4d) in Transition Metal Complexes
The notation "2 5 d 2 4d" is a non-standard, shorthand way that sometimes appears in discussions of transition metal chemistry, particularly when comparing the hybridization schemes of elements from different rows of the periodic table. The "2" likely refers to the involvement of the 2s and 2p orbitals from the central metal atom's valence shell, which are common to both schemes. It essentially contrasts two specific types of octahedral hybridization: one involving the 3d orbitals (often associated with first-row transition metals like Scandium to Zinc) and another involving the 4d orbitals (associated with second-row transition metals like Yttrium to Cadmium). Which means, "2 5 d" can be interpreted as sp³d² hybridization using 3d orbitals, and "2 4d" as sp³d² hybridization using 4d orbitals. This distinction is not merely academic; it profoundly influences the geometry, bond strength, magnetic properties, and reactivity of coordination compounds. This article will demystify this comparison, explaining the fundamental principles of hybridization, the critical differences between using 3d versus 4d orbitals, and why this knowledge is essential for understanding the periodic trends in transition metal chemistry.
Detailed Explanation: The Foundation of Hybridization
To grasp the significance of "2 5 d 2 4d," we must first revisit the concept of orbital hybridization. In simple terms, hybridization is a theoretical model where atomic orbitals (s, p, d) on a central atom mix to form new, equivalent hybrid orbitals. These hybrid orbitals are oriented in specific geometries to maximize separation and minimize electron repulsion, perfectly explaining the observed molecular shapes. For an octahedral geometry—where six ligands are arranged at 90° angles around a central atom—the required hybridization is d²sp³ or sp³d². The order of the letters indicates which orbitals are used first (lower energy) in the mixing process No workaround needed..
The key to understanding the "2 5 d vs 2 4d" comparison lies in quantum numbers and orbital energy. g.The principal quantum number n defines the electron shell (n=1, 2, 3, 4..., 4s and 4p for Period 4 metals), but differ in which d-subshell they employ: 3d for first-row metals or 4d for second-row metals. That said, its available orbitals for bonding are the 4s, 4p, and the 3d orbitals. And ). The "2" might be a misrepresentation of the s/p count (1s+3p=4 orbitals), but the core contrast is 3d vs. Which means this is the "2 5 d" interpretation: using the n=2 s/p? For a first-row transition metal (Period 4), the valence shell is n=4. That's why the 3d orbitals, while belonging to the n=3 shell, are close enough in energy to the 4s and 4p to participate in hybridization. A clearer interpretation is that both schemes use one s and three p orbitals from the same principal quantum level (e.The azimuthal quantum number l defines the subshell type (l=0 for s, l=1 for p, l=2 for d). Wait, no—the "2" is confusing. Still, this yields the d²sp³ scheme, where two 3d, one 4s, and three 4p orbitals combine. 4d participation It's one of those things that adds up..
For a second-row transition metal (Period 5), the valence shell is n=5. So its available orbitals are 5s, 5p, and 4d. This leads to the fundamental difference, therefore, is the principal quantum number of the d-orbitals used in the hybrid set: n-1 d-orbitals for first-row metals (3d for n=4 valence), and n-1 d-orbitals for second-row metals (4d for n=5 valence). Here, hybridization for an octahedral complex involves two 4d, one 5s, and three 5p orbitals, forming d²sp³ hybrids. This is the "2 4d" scheme. This seemingly small change has massive consequences because 4d orbitals are larger, more diffuse, and higher in energy than 3d orbitals.
Step-by-Step Breakdown: Why the D-Orbital Choice Matters
- Orbital Size and Radial Extent: The 4d orbitals have a larger average distance from the nucleus and a more spread-out electron density compared to 3d orbitals. This means a hybrid orbital constructed with a 4d component will inherently be larger and more diffuse than one constructed with a 3d component.
- Overlap with Ligand Orbitals: Effective bonding requires good overlap between the metal's hybrid orbital and the ligand's donor orbital (e.g., a lone pair on NH₃ or CN⁻). The larger 4