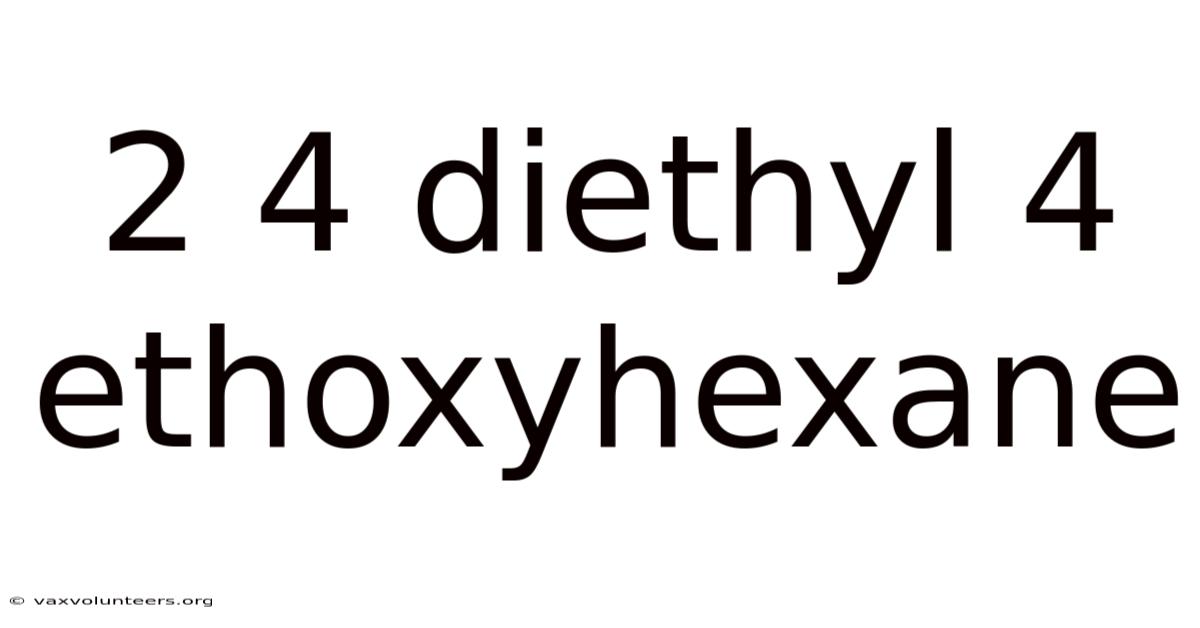Introduction
Organic chemistry relies heavily on a universal language that allows scientists, educators, and students to communicate molecular structures with absolute precision. Because of that, at the heart of this system lies systematic nomenclature, a set of internationally recognized rules designed to eliminate ambiguity. When you encounter a compound name like 2,4-diethyl-4-ethoxyhexane, you are looking at a structured attempt to describe a specific arrangement of carbon, hydrogen, and oxygen atoms. Understanding how to decode such names is not merely an academic exercise; it is a foundational skill that bridges theoretical chemistry with practical laboratory work, pharmaceutical development, and materials science.
This article explores the meaning, structure, and educational significance of 2,4-diethyl-4-ethoxyhexane in depth. While the name appears straightforward at first glance, it actually serves as a classic teaching tool used to highlight common pitfalls in chemical naming conventions. On top of that, by breaking down each component of the name, examining the underlying structural implications, and applying official IUPAC guidelines, you will gain a comprehensive understanding of why systematic nomenclature matters and how to approach complex organic compounds with confidence. Whether you are a beginner navigating your first chemistry course or an experienced learner refining your analytical skills, this guide will provide a complete, structured pathway to mastering the concept Small thing, real impact..
Detailed Explanation
To truly grasp what 2,4-diethyl-4-ethoxyhexane represents, we must first understand how organic compounds are named. The prefix diethyl signals the presence of two ethyl groups, each consisting of two carbon atoms attached to the main chain. Also, in this case, the parent chain is hexane, indicating a continuous backbone of six carbon atoms. Practically speaking, the name is constructed using the International Union of Pure and Applied Chemistry (IUPAC) framework, which breaks down molecular structures into three primary components: a parent chain, substituent groups, and numerical locants. Meanwhile, ethoxy refers to an oxygen-containing functional group, specifically an ether linkage where an ethyl group is bonded to an oxygen atom.
The numbers 2 and 4 are locants that specify exactly where these substituents attach to the hexane backbone. And according to the name, one ethyl group is bonded to the second carbon, while both the second ethyl group and the ethoxy group are attached to the fourth carbon. At first glance, this seems like a perfectly valid description. Still, organic chemistry naming is governed by strict hierarchical rules that prioritize the longest continuous carbon chain, the lowest possible numbering for substituents, and proper alphabetical ordering. When we apply these rules rigorously, the name 2,4-diethyl-4-ethoxyhexane reveals structural contradictions that make it chemically inaccurate under official standards.
Understanding this compound requires more than memorizing prefixes and numbers; it demands a working knowledge of molecular geometry and chain identification. The presence of branched alkyl groups and an ether functional group creates a three-dimensional structure that influences physical properties like boiling point, solubility, and reactivity. Ethers, in particular, are characterized by an oxygen atom bridging two carbon chains, which imparts unique chemical behavior compared to pure hydrocarbons. Recognizing how these functional groups interact with the parent chain is essential for predicting how the molecule will behave in real-world chemical environments, from industrial solvents to biological systems It's one of those things that adds up..
Step-by-Step or Concept Breakdown
Decoding a systematic chemical name requires a methodical approach that transforms text into a visual molecular structure. The first step involves identifying the parent hydrocarbon, which in this case is hexane. You begin by drawing a straight chain of six carbon atoms and numbering them sequentially from left to right. This numbered backbone serves as the foundation upon which all substituents will be attached. It is crucial to remember that the numbering direction is not arbitrary; it must eventually align with IUPAC rules that prioritize the lowest possible locant numbers for all branches and functional groups And it works..
The second step focuses on placing the substituent groups at their designated positions. The prefix diethyl tells us to attach two ethyl groups, which are two-carbon alkyl chains. One ethyl group goes on carbon number two, and the second goes on carbon number four. Next, we address the ethoxy group, which consists of an oxygen atom bonded to an ethyl chain. This group is also placed on carbon number four, meaning that the fourth carbon becomes a tertiary center bonded to three different carbon chains and one oxygen atom. At this stage, you have successfully translated the written name into a skeletal structure, but the work is not yet complete.
The final and most critical step involves verifying the longest continuous carbon chain. Once the longest chain is identified, the substituents must be renumbered to ensure the lowest possible locants, and the name must be reconstructed accordingly. When you trace through the structure implied by 2,4-diethyl-4-ethoxyhexane, you will discover that incorporating one of the ethyl branches into the main backbone actually creates a seven-carbon chain. This means the true parent hydrocarbon should be heptane, not hexane. IUPAC rules mandate that the parent chain must be the longest possible sequence of carbon atoms, regardless of how the molecule was initially drawn. This step-by-step verification process is what transforms a superficial label into an accurate chemical identity.
Real Examples
In academic and laboratory settings, 2,4-diethyl-4-ethoxyhexane frequently appears as a deliberate naming exercise rather than a commercially available compound. University chemistry courses routinely include intentionally flawed names in exams and problem sets to test students' ability to apply IUPAC rules critically. Still, for instance, a professor might present this name and ask students to draw the structure, identify the longest chain, and provide the correct systematic name. This pedagogical approach forces learners to move beyond rote memorization and develop analytical reasoning skills that are essential for advanced organic synthesis, spectroscopy interpretation, and molecular modeling Simple, but easy to overlook..
Beyond the classroom, the principles demonstrated by this compound have direct applications in pharmaceutical development and materials engineering. So ethers and branched alkanes are foundational components in solvent systems, polymer precursors, and drug delivery molecules. Understanding how to correctly name and classify these structures ensures that researchers can communicate safely and accurately about chemical handling, toxicity profiles, and reaction pathways. A misnamed compound in a research paper or safety data sheet could lead to dangerous laboratory errors, highlighting why precise nomenclature is not just an academic formality but a critical professional standard Most people skip this — try not to..
Scientific or Theoretical Perspective
The theoretical foundation of chemical naming rests on the IUPAC nomenclature system, which was established to create a universal, unambiguous language for chemistry. Still, the system operates on several core principles: selecting the longest continuous carbon chain as the parent, identifying all functional groups and substituents, assigning the lowest possible locant numbers, and arranging prefixes alphabetically. Day to day, when multiple substituents occupy the same carbon, as seen with the ethyl and ethoxy groups on carbon four, the molecule becomes a branched tertiary center with specific stereochemical and electronic implications. The oxygen atom in the ethoxy group introduces polarity, altering intermolecular forces compared to a purely hydrocarbon structure.
From a molecular orbital perspective, the presence of an ether linkage affects electron distribution across the molecule. Oxygen is highly electronegative, meaning it pulls electron density away from adjacent carbon atoms, creating a partial negative charge on the oxygen and partial positive charges on neighboring carbons. On the flip side, this dipole moment influences how the molecule interacts with solvents, participates in nucleophilic substitution reactions, and aligns in crystalline or liquid phases. Theoretical chemistry also examines how branching affects thermodynamic stability; highly branched alkanes typically exhibit lower boiling points than their straight-chain isomers due to reduced surface area and weaker London dispersion forces Took long enough..
Common Mistakes or Misunderstandings
One of the most frequent misconceptions surrounding 2,4-diethyl-4-ethoxyhexane is the assumption that the name is inherently correct. Plus, many students accept the provided label at face value without questioning whether it adheres to IUPAC hierarchy. This passive approach leads to structural misinterpretations, incorrect reaction predictions, and poor performance in advanced chemistry assessments. The reality is that the name violates the fundamental rule of longest-chain identification, making it a textbook example of how easily nomenclature errors can propagate when critical verification steps are skipped That alone is useful..
Another common misunderstanding involves confusing ethoxy with ethyl. While both prefixes contain the root "eth," they represent entirely different chemical entities. Ethyl is a two-carbon alkyl group, whereas ethoxy is a two-carbon chain attached to an oxygen atom, forming an ether functional group