1s2 2s2 2p6 3s2 3p4
vaxvolunteers
Mar 16, 2026 · 4 min read
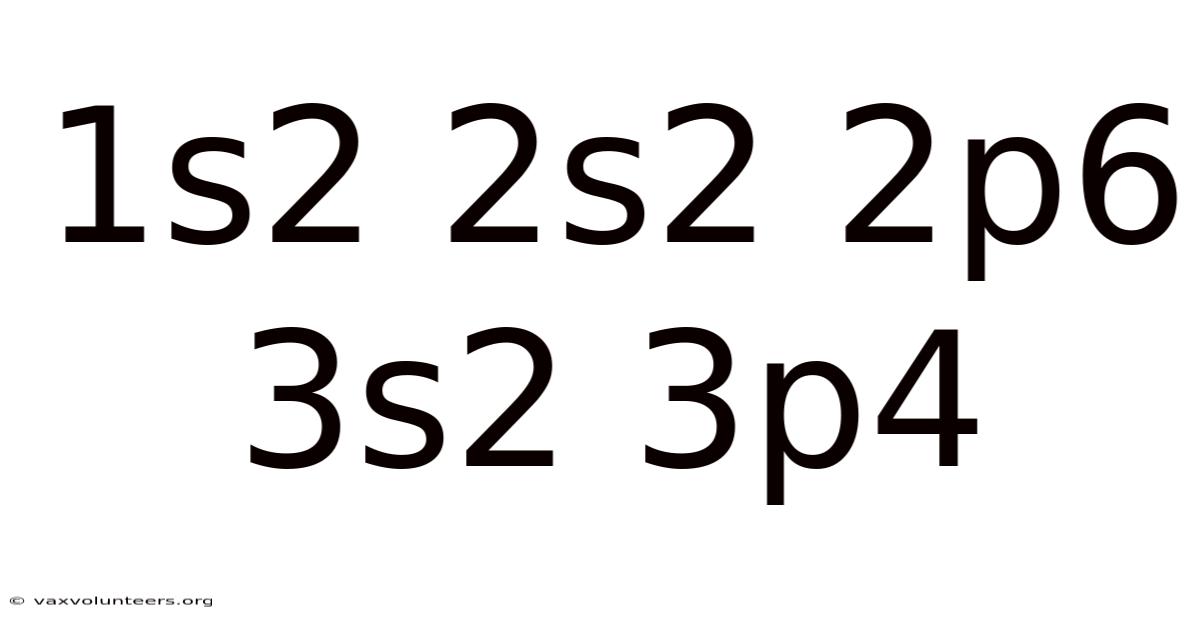
Table of Contents
Understanding the Atomic Blueprint: Decoding 1s² 2s² 2p⁶ 3s² 3p⁴
At first glance, the string 1s² 2s² 2p⁶ 3s² 3p⁴ looks like a cryptic code from a science fiction film. In reality, it is one of the most fundamental and powerful pieces of information in chemistry and physics: the electron configuration of a specific element. This precise notation is the atomic blueprint for the element sulfur (S), atomic number 16. It describes exactly how its 16 electrons are distributed among the various atomic orbitals surrounding the nucleus. Mastering this notation is akin to learning the alphabet of the periodic table; it unlocks the reasoning behind an element's chemical personality, its reactivity, its place in the table, and its ability to form the incredible diversity of molecules that constitute our world. This article will serve as a comprehensive guide to decoding this configuration, transforming that cryptic string into a clear and meaningful story about the sulfur atom.
Detailed Explanation: The Language of Electrons
To understand 1s² 2s² 2p⁶ 3s² 3p⁴, we must first understand the components of the notation itself. The system is based on the quantum mechanical model of the atom, which supersedes the simpler Bohr model.
- The Principal Quantum Number (n): The number at the beginning (
1,2,3) indicates the principal energy level or shell. Think of these as the major "neighborhoods" or "floors" of the electron cloud, withn=1being closest to the nucleus and lowest in energy,n=2being the next, and so on. The energy and average distance from the nucleus increase withn. - The Subshell Letter (s, p, d, f): The letter indicates the subshell or orbital type within that energy level. Each subshell has a characteristic shape defined by its angular momentum quantum number:
- s subshells are spherical.
- p subshells have a dumbbell shape (three orbitals, oriented along x, y, z axes).
- d and f subshells have more complex shapes (five and seven orbitals, respectively).
- The Superscript Number: The number written as a superscript (
²,⁶,⁴) tells us exactly how many electrons occupy that specific subshell.
The order in which these subshells are filled is not simply 1, 2, 3 but follows a specific sequence dictated by their relative energies, often remembered by the Aufbau principle (from the German aufbau, meaning "building up"). The standard order is: 1s → 2s → 2p → 3s → 3p → 4s → 3d → 4p... This order is derived from solving the Schrödinger equation for multi-electron atoms.
Now, let's read our sulfur configuration: 1s² 2s² 2p⁶ 3s² 3p⁴.
1s²: The first two electrons go into the lowest energy orbital, the 1s spherical orbital.2s²: The next two fill the 2s orbital.2p⁶: The following six electrons completely fill the three 2p orbitals (each can hold 2 electrons, 3 orbitals x 2 = 6).3s²: The next two electrons move out to the 3s orbital.3p⁴: The final four electrons (for sulfur's 16 total) begin to fill the three 3p orbitals. According to Hund's rule, which states that electrons will occupy degenerate orbitals (orbitals of the same energy) singly before pairing up, these four electrons will be arranged as: three electrons each in a separate 3p orbital (with parallel spins), and the fourth electron will pair up in one of those orbitals. This gives sulfur a configuration of[↑↓] [↑] [↑]for its 3p subshell.
This distribution results in a core of 1s² 2s² 2p⁶ (10 electrons, identical to the noble gas neon) and a valence shell of 3s² 3p⁴ (6 electrons). The valence electrons are the outermost electrons and are primarily responsible for an element's chemical bonding behavior.
Step-by-Step Breakdown: Building the Sulfur Atom
Let's construct the sulfur atom's electron configuration systematically, applying the three fundamental rules of electron assignment.
- Start at the Bottom (Lowest Energy): The 1s orbital is the lowest energy. Place 2 electrons here:
1s². (Electrons 1 & 2). - Fill the Next Lowest: The 2s orbital is next. Place 2 electrons:
2s². (Electrons 3 & 4). - Fill the 2p Subshell: The three 2p orbitals are the next lowest and are degenerate (same energy). According to Hund's rule, we place one electron in each orbital before pairing.
- Electron 5 goes into 2pₓ:
2p¹ - Electron 6 goes into 2pᵧ:
2p² - Electron 7 goes into 2p_z:
2p³ - Now we pair up. Electron 8 pairs in 2pₓ:
2p⁴ - Electron 9 pairs in 2pᵧ:
2p⁵ - Electron 10 pairs in 2p_z:
2p⁶. The 2p subshell is now full.
- Electron 5 goes into 2pₓ:
- Move to the Third Shell: The next lowest available orbital is 3s. Place 2 electrons:
3s². (Electrons 11 &
Latest Posts
Latest Posts
-
38 Degrees F To C
Mar 16, 2026
-
52 Thousandths In Scientific Notation
Mar 16, 2026
-
How Many Inches Is 7ft
Mar 16, 2026
-
Actual Dimensions Of A 2x6
Mar 16, 2026
-
Alcohol In Any Concentration Is
Mar 16, 2026
Related Post
Thank you for visiting our website which covers about 1s2 2s2 2p6 3s2 3p4 . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.