1 3 X 10 4
vaxvolunteers
Mar 09, 2026 · 4 min read
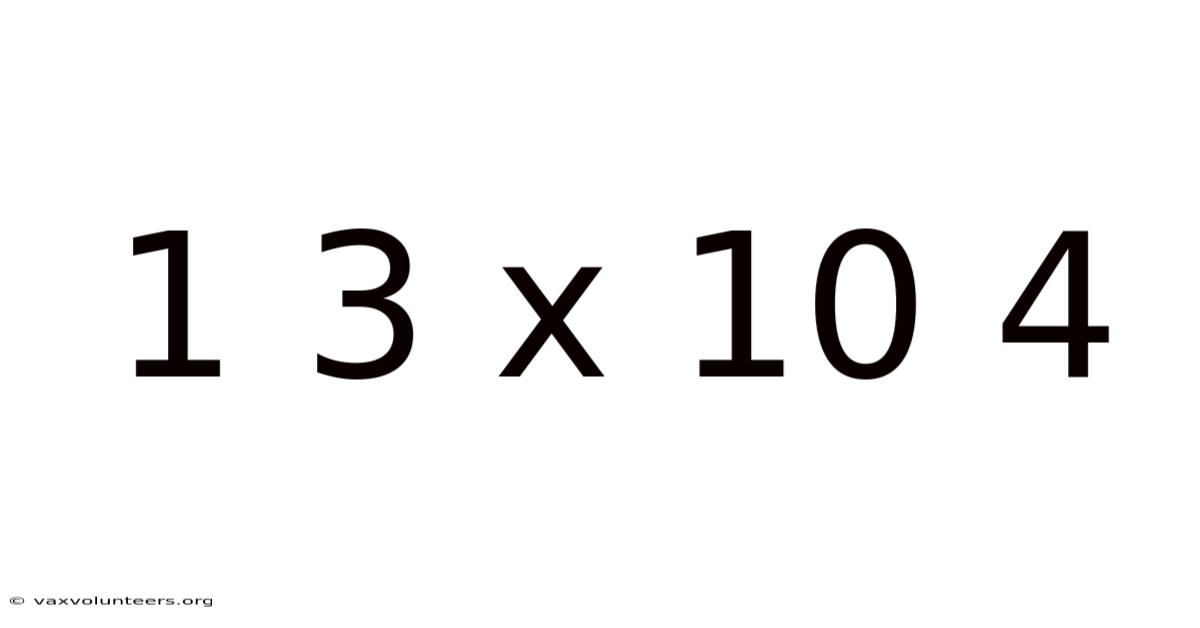
Table of Contents
Understanding 1.3 × 10⁴: A Deep Dive into Scientific Notation
Have you ever tried to write the distance from the Earth to the Sun in standard decimal form? You’d be meticulously writing 149,600,000, and that’s just for one astronomical unit! Dealing with such colossal numbers—or incredibly tiny ones, like the mass of a proton—quickly becomes unwieldy. This is where a powerful mathematical tool comes to the rescue: scientific notation. The expression 1.3 × 10⁴ is a perfect, compact example of this system. At first glance, it might seem like a cryptic code, but it’s simply an elegant way to represent the number 13,000. This article will unpack everything about this notation, from its fundamental structure to its profound applications in science and engineering, ensuring you not only understand how to read it but also why it is an indispensable part of modern quantitative literacy.
Detailed Explanation: The Anatomy of a Number
Scientific notation is a standardized method for writing numbers that are either extremely large or extremely small. Its core purpose is to simplify representation and calculation, reducing the chance of error when counting dozens of zeros. The general form is a × 10ⁿ, where:
- a is the coefficient (or mantissa), a number greater than or equal to 1 and less than 10 (1 ≤ |a| < 10). In our example, a = 1.3.
- 10ⁿ is the base raised to an integer exponent (n). The exponent tells us how many places to move the decimal point to convert back to standard form.
The genius of this system lies in its separation of a number’s magnitude (handled by the exponent) from its significant figures (handled by the coefficient). The exponent 4 in 1.3 × 10⁴ is positive, indicating a large number. It means we take the coefficient, 1.3, and move its decimal point 4 places to the right. If the exponent were negative (e.g., 1.3 × 10⁻⁴), we would move the decimal point to the left, representing a tiny fraction.
This notation is not arbitrary; it is deeply connected to our base-10 number system. Each single-place move of the decimal point corresponds to a multiplication or division by 10. Therefore,
an exponent of 4 means multiplying by 10 four times, which is exactly 10⁴ = 10,000. Multiplying 1.3 by 10,000 gives 13,000, confirming the equivalence.
Practical Applications: Why It Matters
The true power of scientific notation becomes evident in real-world applications. In physics, the speed of light is approximately 3 × 10⁸ meters per second—far more manageable than writing 300,000,000. In chemistry, Avogadro's number, the count of atoms in a mole, is 6.022 × 10²³, a number so vast it defies intuitive comprehension without this shorthand. Even in biology, the diameter of a DNA helix is about 2 × 10⁻⁹ meters, a scale only expressible concisely through scientific notation.
Engineering and technology rely on it just as heavily. Microprocessors operate at frequencies like 3.5 × 10⁹ Hz (3.5 GHz), and data storage is measured in terabytes (10¹² bytes). Without scientific notation, calculations in these fields would be riddled with errors and inefficiencies.
Calculations and Comparisons
Working with numbers in scientific notation streamlines arithmetic. To multiply 1.3 × 10⁴ by 2 × 10³, you multiply the coefficients (1.3 × 2 = 2.6) and add the exponents (10⁴ × 10³ = 10⁷), yielding 2.6 × 10⁷. This is far quicker than multiplying 13,000 by 2,000 in standard form. Similarly, dividing 1.3 × 10⁴ by 1.3 × 10² gives 1 × 10², or 100, with minimal effort.
Comparisons are also more intuitive. A number with a higher exponent is always larger, regardless of the coefficient. Thus, 1.3 × 10⁴ is ten times greater than 1.3 × 10³, a relationship immediately clear from the exponents.
Conclusion
Scientific notation, as exemplified by 1.3 × 10⁴, is far more than a mathematical convenience—it is a universal language for expressing the extremes of scale that define our universe. From the subatomic to the cosmic, it allows scientists, engineers, and students to communicate and compute with clarity and precision. Understanding its structure and logic not only demystifies intimidating numbers but also opens the door to deeper engagement with the quantitative world. Whether you're calculating astronomical distances or microscopic measurements, scientific notation is an essential tool, transforming the unmanageable into the understandable.
Latest Posts
Latest Posts
-
Who Primarily Used A Stoa
Mar 10, 2026
-
Ballistic Stretching Is Best Performed
Mar 10, 2026
-
In General Ai Bi
Mar 10, 2026
-
Convert 185 Pounds To Kg
Mar 10, 2026
-
Intentional Withholding Of Material Facts
Mar 10, 2026
Related Post
Thank you for visiting our website which covers about 1 3 X 10 4 . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.